Tranexamic acid use in the non-surgical neonatal population: a scoping review
Tranexamic acid (TXA) is recommended for use in paediatric major haemorrhage situations and paediatric surgery where major blood loss is anticipated. There are no recommendations regarding the use of TXA for major bleeding for neonates in a non-surgical context. This review evaluates existing literature relating to TXA use in neonates outside the surgical context. The limited available evidence indicates TXA can be a useful adjunct in management of neonatal non-surgical haemorrhage.
Jennifer Peterson1,2Neonatal Sub-Speciality Trainee
jennifer.peterson@hotmail.co.uk
Kate Pritchard3
Transport Advanced Neonatal Nurse Practitioner
Susan Kamupira2
Consultant Neonatologist
Ruth Gottstein2
Consultant Neonatologist
Ian Dady3
Consultant Neonatologist
1Faculty of Biology, Medicine and Health, University of Manchester
2Neonatal Intensive Care Unit, St Mary’s Hospital, Manchester
3Connect North West Neonatal Transport Team, St Mary’s Hospital, Manchester
Peterson J., Pritchard K., Kamupira S., Gottstein R., Dady I. Tranexamic acid use in the non-surgical neonatal population: a scoping review. Infant 2024; 20(2): 49-54.
Tranexamic acid (TXA) is a synthetic lysine analogue, administered for its antifibrinolytic effects. It is commonly used in paediatric trauma protocols and cardiac surgery with the intention of minimising bleeding and blood loss.1-3 Dosing regimens have not been well researched in paediatric or neonatal populations and are instead extrapolated from adult dosing regimens.4-8 Administration carries the risk of significant side-effects such as dose-dependent seizures, which have been reported after cardiac surgery.4,9 The utility of TXA is infrequently reported in the non-surgical neonatal population. However, in instances of excessive or prolonged bleeding, TXA may have a role in aiding bleeding cessation in neonates.
This scoping review evaluates the reported cases of non-surgical TXA use in neonates to date, summarising the experience, dosing, indications and outcomes of TXA use in the newborn population.
Methods
Search strategy
We evaluated the existing literature relating to TXA use in neonates outside the surgical context. The Cochrane Reviews, Web of Science and PubMed databases were searched. Two researchers (JP and KP) independently reviewed the search results and then screened the titles and abstracts. A third researcher (ID) was available to review and resolve any inconsistencies. Full text versions of potentially eligible studies were sourced for review. The following search strategy was used: (‘neonat*’) AND (‘tranexamic acid’). The search was conducted in September 2023 and there were no date range restrictions imposed. This study is based exclusively on published literature and therefore did not require formal ethics panel review.
Criteria for inclusion in this review
The inclusion and exclusion criteria were agreed by the authors prior to starting the review. Articles describing TXA use in neonates in non-surgical settings were included. In vivo/in vitro results pertaining to neonates or paediatrics were considered eligible. Articles describing TXA use in adult populations were excluded. Articles describing TXA use in obstetric, paediatric patients (>1 month of age), paediatric cardiac surgery or paediatric general surgery were excluded. Articles written in any language other than English were excluded.
Data collection
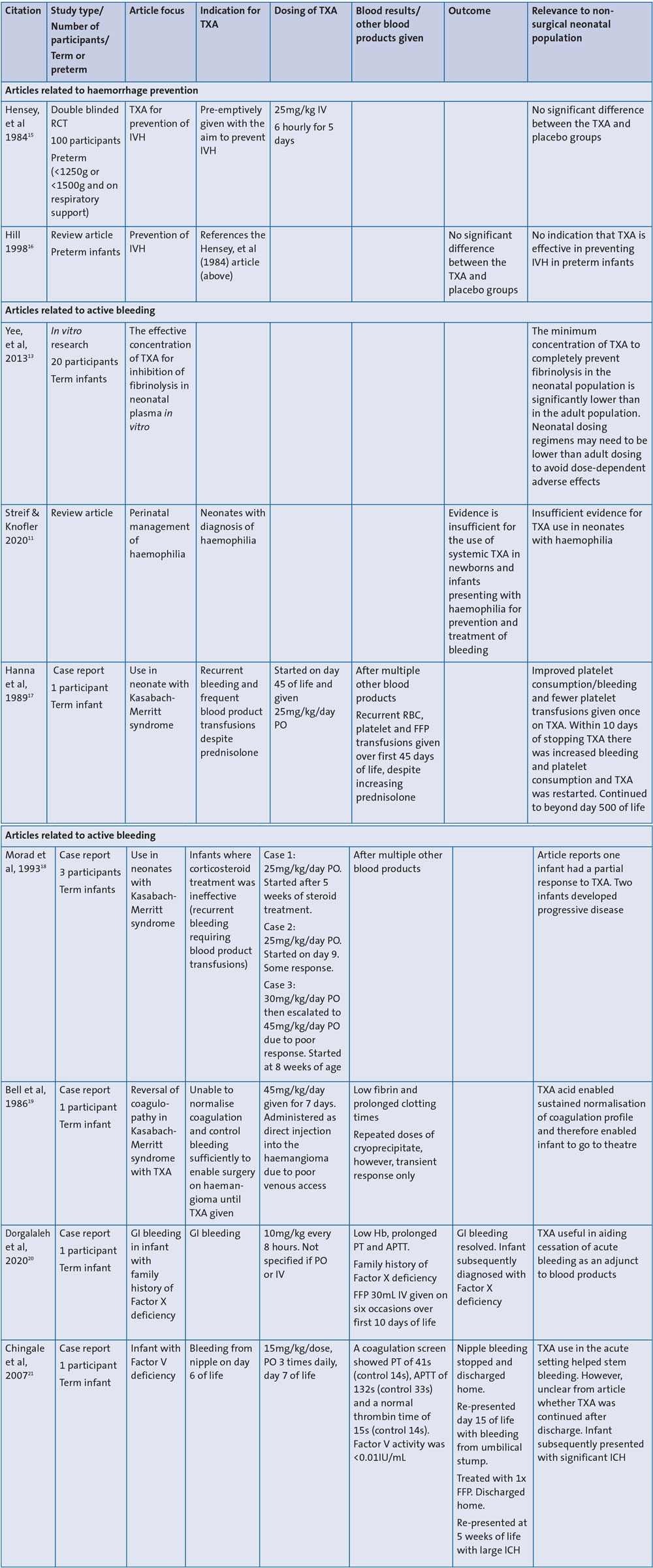
Two researchers (JP and KP) independently extracted key data from the included studies. A third researcher (ID) checked the extracted data. For each study, the authors retrieved relevant data on study type and characteristics, demographics of included patients, diagnoses and indication for TXA use, laboratory data, additional blood products provided and outcome. The authors summarised the relevance of each paper to the systematic review intention, which is to evaluate non-surgical TXA use in neonates (TABLE 1).
TABLE 1 A summary of the included articles. Key: TXA=tranexamic acid; RCT: randomised controlled trial; IVH=intraventricular haemorrhage; PO=per oral; RBC=red blood cell; FFP=fresh frozen plasma; GI=gastrointestinal; IV=intravenous; Hb=haemoglobin; PT=partial thromboplastin; APTT=activated partial thromboplastin time; ICH=intracranial haemorrhage.
Quality appraisal of included studies
The search strategy resulted in nine articles that were included in this scoping review. There was significant heterogeneity between the included studies and this limited synthesis of the results. Due to the variation in article type and that the majority were case reports, the Murad, et al tool10 was utilised for evaluating the quality of included studies.
The quality of the available data for TXA use in non-surgical neonates was poor (FIGURE 1). This was primarily due to several articles concerning selective use of TXA in specific syndromes, such as Kasabach-Merritt syndrome. Quality of selection was overall low (78%) with the majority of articles selectively reporting individual or small numbers of infants rather than reflecting the wider population of that condition or population. Quality was low for causality (100%), particularly in allowing for alternative causes of bleeding cessation. Many included articles reported TXA had been administered alongside multiple other blood products with no comment regarding the specific effect noted after the TXA administration.
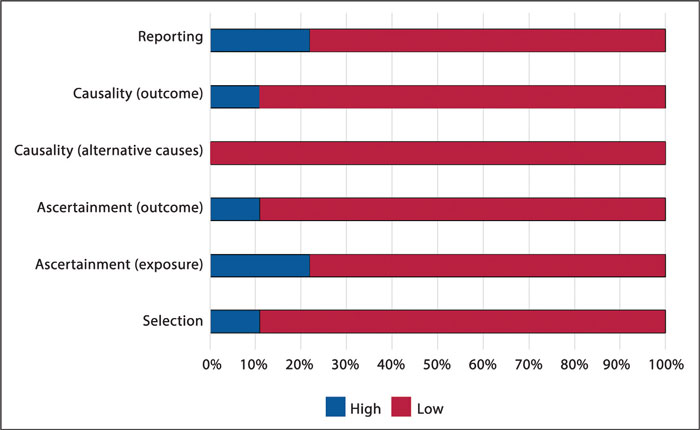
FIGURE 1 A summary of the quality assessment of the included studies.
Data synthesis
A narrative synthesis of included studies was conducted as the nature of the included studies does not allow a meaningful meta-analysis to be performed.
Results
There were nil applicable returns from the Web of Science (nil results) and Cochrane Reviews databases (one result reviewing TXA use in postpartum haemorrhage). The study search strategy for PubMed yielded 129 non-duplicated articles. Following a manual review, 109 were excluded because the article related to TXA use in either adult, obstetric or general paediatric and/or cardiac surgery, and two that were only available in Italian. Of the remaining 20 articles, 11 articles were excluded following review of the full article as the TXA use described related to either adult or non-neonatal paediatric patients (FIGURE 2).
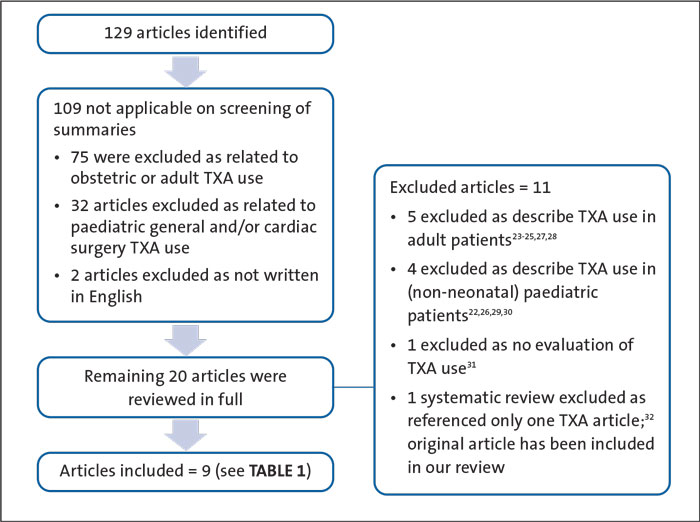
FIGURE 2 A flow chart of the included studies. The included articles can be seen in TABLE 1.
Discussion
This scoping review shows a paucity of reported use of non-surgical TXA in the neonatal population. The included articles have significant heterogeneity in age, indication, dosing regime and route of administration (TABLE 1).
Indications for use
There was significant variety in the indications for non-surgical use of TXA in neonates in the reported literature (TABLE 2).

TABLE 2 The literature-reported indications for non-surgical use of TXA in neonates. Key: IVH=intraventricular haemorrhage.
Haemorrhage prevention
Of the articles identified by this review, three reported on use of TXA to attempt to prevent haemorrhage in neonates. Two of the three articles discussed use of TXA in prevention of intraventricular haemorrhage (IVH) in premature infants and one article discussed TXA use in prevention of haemophilia-related bleeding. These articles found no evidence that administration of TXA can prevent IVH or haemophilia-related bleeds in neonates.
Active treatment of bleeding
The remainder of the included articles focused on administration of TXA to reduce active haemorrhage in neonates. There remains variation in the specific presentation of the bleeding (bleeding from haemangioma, bleeding from nipple, bleeding from GI tract, TABLE 1). While the article reviewing perinatal haemophilia management11 concluded there was insufficient evidence to recommend TXA use for prevention or treatment of haemophilia bleeds in neonates, the other included articles (chiefly case reports regarding use in excessive bleeding from haemangiomas) report overall positive responses to TXA. The five case report articles included in this review, report a reduction or cessation of bleeding following administration of TXA.
Dosing in the neonatal population
The dosing regimen used was reported in six of the nine articles. Of these six, in five the indication of use was to manage active bleeding and one was for IVH prevention. The IVH prevention regimen used TXA at 100mg/kg/day (25mg/kg six-hourly) IV for five days. This prophylactic regimen did not show a reduction of IVH risk.
Dosing regimens for active bleeding
The remaining five articles were all small-scale case reports reporting on TXA use in active bleeding. Across these five articles the dosing regimens for seven infants were provided. The majority of infants received TXA orally (five infants). There was one case report describing highly unusual TXA administration via direct injection into a haemangioma. This was reportedly due to difficult IV access. In the final case, the route of TXA administration was not specified. Dosing regimens varied widely from 25mg/kg/day up to 45mg/kg/day (TABLE 3). The reported regimes are consistent with the recommendations from the British National Formulary for Children.12 Of note, as with many medications used in neonatal medicine, TXA is not licensed in children under the age of one year.
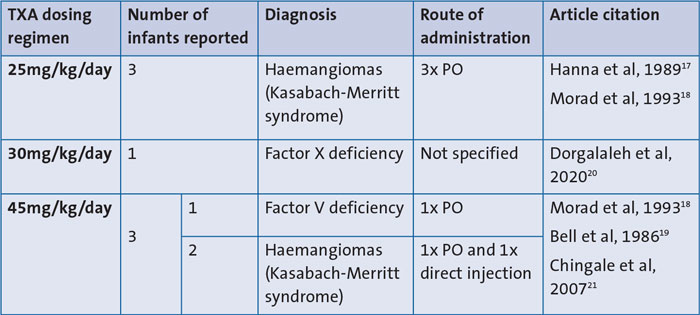
TABLE 3 Literature reported dosing regimens for TXA use in non-surgical neonates. Key: PO=per oral.
The majority of included articles did not provide the rationale for the selected dosing regimen. In one case describing TXA use in haemangiomas, the authors specified that TXA had been started at 30mg/kg/day and then increased up to 45mg/kg/day due to ongoing bleeding. The variation in TXA dosing reflects the lack of evidence-based guidance for dosing regimens in the neonatal population.
TXA carries a risk of side-effects that is dose dependent. Side-effects can be significant with the potential for infants to develop seizures, hypotension, embolism and thrombosis.12 Of note, TXA is renally excreted and therefore, there is an increased risk of accumulation and consequent increased risk of side-effects in infants with any degree of renal impairment.12
In summary
TXA has become a core component of adult and paediatric trauma management pathways and there is a good evidence base for its use in these situations. The results of this scoping review show that there is limited reported experience of TXA use in the non-surgical neonatal population. There were 129 articles identified with 120 of these excluded as, on review, they did not pertain to neonatal non-surgical TXA use.
From the current available evidence, it appears that TXA has no role in the prevention of haemorrhage in neonates. However, there is some evidence, albeit limited, that TXA can serve as a useful adjunct in management of non-surgical haemorrhage in neonates with underlying medical pathology. Future research should actively focus on TXA use in this context.
There is limited evidence for current dosing regimens in neonates, who have altered pharmacokinetics when compared to adults and may be at higher risk of TXA dose-dependent adverse events. Further research is needed to investigate the optimal dosing regimen for TXA in neonatal patients. As the in vitro study conducted by Yee et al13 illustrates, the neonatal response to TXA varies from adults, with neonates requiring a significantly lower concentration of TXA to inhibit fibrinolysis compared to adult populations. Dosing regimens derived from adult and paediatric protocols may therefore, recommend doses of TXA that are excessive for a neonatal patient. This was clinically demonstrated by Wesley et al14 who profiled plasma TXA concentrations across different paediatric age groups. The patients included in the Wesley study were all undergoing cardiopulmonary bypass surgery, which involves deep hypothermic circulatory arrest and ultrafiltration and is therefore, markedly different to the population focus of this review. Of note, Wesley, et al did demonstrate a significantly different TXA plasma concentration profile in the <12-month age group and highlight the need for reduced dosing regimens in neonatal patients.
Given that the complications of TXA occur in a dose-dependent manner, it is particularly important that doses exceeding the minimum required for the beneficial effect should be avoided wherever possible. Future research is needed to identify optimal use and dosing regimens for neonatal patients with major haemorrhage from underlying medical pathologies.
Author contributions
ID devised the project. JP, KP and ID reviewed the data. JP and KP wrote the manuscript. SK, RG and ID supervised the project and edited the manuscript. All authors reviewed the manuscript prior to submission.
Or read this article in our
Tablet/iPad edition
- This literature review looks at TXA use for major haemorrhage in neonates in the non-surgical context.
- TXA may be a useful adjunct for major haemorrhage in neonates with medical pathology.
- There is risk of dose-dependent adverse events with current TXA dosing regimens.
Also published in Infant: