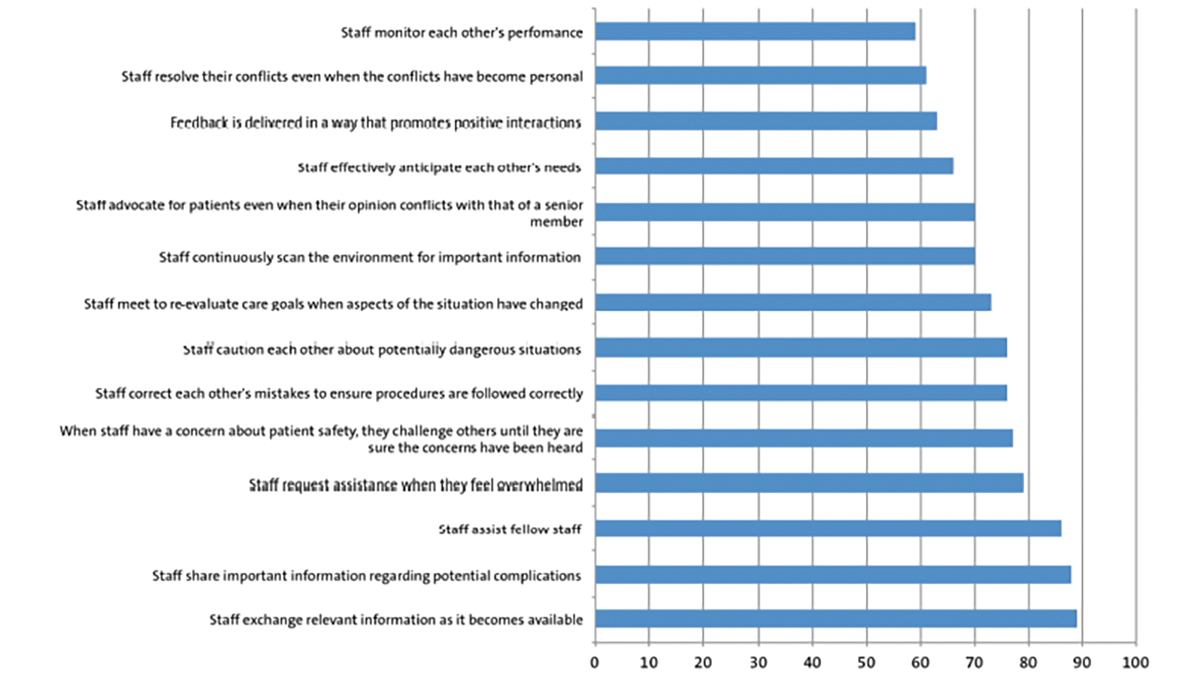
Congenital myofibroma: an unusual neonatal presentation
The diagnosis of a vascular cutaneous appearing lesion in a neonate can be challenging. We report a case of a solitary congenital myofibroma presenting at birth as a major bleeding lesion. Further to the major haemorrhage and the ensuing hypoxia/ischaemia, the baby suffered multi-organ failure. This eventually led to redirection of care and neonatal death in about two weeks. On histopathological examination, the lesion showed a pattern identified as haemangio-pericytoma. This is a rare tumour involving blood vessels and soft tissues. In our literature search there is no other case report of a myofibromatous lesion presenting in this way. The importance of having an emergency haemorrhage protocol was highlighted with this incident.
Ajinkya Kale1
MTI Fellow in Neonates
Thè Thwin1
Consultant Neonatologist
Srinivasarao Babarao2
Consultant Neonatologist
srinivasarao.babarao@lwh.nhs.uk
Rajeev Shukla3
Consultant Paediatric Pathologist
1Wirral University Teaching Hospital NHS Foundation Trust
2Liverpool Women’s Hospital
3Alder Hey Children’s Hospital, Liverpool
The case
A 32-year-old pregnant woman was admitted to the hospital due to reduced fetal movements and pinkish vaginal discharge for three days. She had a normal antenatal period, including the 20-week anomaly scan.
At presentation at 32 weeks of pregnancy her clinical condition was stable and her membranes were intact. Her initial and continuous cardiotocograph (CTG) monitoring was within normal limits.
She received two doses of steroids in anticipation of preterm labour. After approximately 30 hours of admission, the CTG became pathological and an emergency caesarean section was performed. During the caesarean section there was some difficulty in extraction of the baby. A large, bleeding mass attached to the baby’s left upper limb was found to be the cause of this difficulty. The baby was born in poor condition: pale, floppy, not making any respiratory effort and was bleeding from the lesion. Initially, it was difficult to see the origin of the lesion, as it lay to the left of the baby’s head. The position of the lesion made it difficult to use the facemask while resuscitating the baby. The baby needed full resuscitation including intubation, cardiac compressions, and drugs/blood transfusion through an umbilical venous line. Amid the resuscitation, the origin of the exsanguinating lesion was identified to be from the left elbow (FIGURE 1).
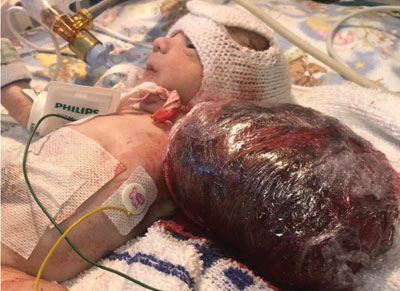
FIGURE 1 The large bleeding lesion presenting at birth.
A cord tie was fixed around the stump. The mass was wrapped tightly with cling film. This seemed to stop the bleeding transiently. The cord haemoglobin was noted to be 60gm/L, indicating that the lesion had started to bleed in utero.
After admission to the neonatal unit the baby continued to bleed from the lesion and needed further resuscitation with blood and blood products. The tourniquet was re-tightened, with minimal effect on bleeding. Due to haemodynamic instability, the baby was commenced on inotropic support. Also, tranexamic acid and additional vitamin K were administered. Another tourniquet was placed on the left axillary area in an attempt to reduce the bleeding. Blood investigations showed a significantly deranged coagulation profile, indicative of disseminated intravascular coagulation. The regional neonatal transport team was contacted and the baby was urgently transferred to the regional quaternary children’s hospital for surgical intervention. The vascular lesion was removed maximally. It was not possible to perform fasciotomy due to ongoing haemodynamic instability and the deranged coagulation profile. The baby deteriorated further after admission to the intensive care unit and suffered multi-organ failure, including necrotising enterocolitis. He was managed with mechanical ventilation, haemodialysis and repeated blood transfusions. Care was redirected to palliation after discussion with the parents, due to poor clinical condition and prospects of adverse prognosis.
Pathological examination of the lesion showed the nodular mass to weigh about 470g and measuring approximately 125x115x57mm. The surface of the lesion was extensively ulcerated and haemorrhagic. The cut surface was also variegated, soft and extensively haemorrhagic, with no obvious necrosis (FIGURE 2).
Microscopically, the lesion showed a rich vascular network comprising thin-walled vessels, some of which had haemangio-pericytomatous appearance (FIGURE 3). The morphological appearances were consistent with myofibroma.
Discussion
Infantile myofibromas (IM), although rare, are the most common fibrous tumours seen in infants. They are often present at birth (75% of cases) and 90% of cases appear within the first two years of life. The clinical presentation is variable. Involvement of extremities is a rare manifestation: a solitary IM on the upper extremity accounts for only 13.3% of cases. IMs may be solitary (70-80%) or multi-centric (20-30%). The solitary form is the most common with a male predominance.
The most common locations of a solitary IM are the head and neck, followed by the trunk and extremities. They present as non-tender, rubbery, subcutaneous or dermal nodules of 0.5-7cm in diameter that are dusky-red to purple in colour. Surface telangiectasias may be noted and ulceration occurs rarely. Their appearance frequently leads to confusion in distinguishing them from congenital haemangiomas. The multicentric form of IM can present with a few to up to 100 lesions and occasionally a large lesion is surrounded by multiple smaller lesions. Approximately one third of multicentric myofibromas have visceral involvement.
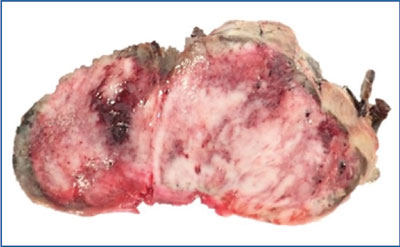
FIGURE 2 Pathological examination of the lesion (125x115x57mm, 470g) showing extensive ulceration and haemorrhage.
FIGURE 3 Microscopically, the lesion showed a rich vascular network comprising thin-walled vessels, some of which had haemangiopericytomatous appearance.
Most cases of IM are thought to be sporadic. Familial forms have been reported with autosomal dominant and recessive inheritance patterns. Mutations in the PDGFRB (platelet-derived growth factor receptor beta) and NOTCH3 gene have been identified in the autosomal dominant forms of the disease. Genetic counselling should be considered in familial cases, as future offspring may be affected.
Histopathology helps in providing a definitive diagnosis. IMs have a characteristic histological pattern with an outer zone of spindle-shaped myofibroblasts arranged in fascicles and an inner zone of round cells with enlarged hyperchromatic nuclei surrounding thin walled haemangiopericytoma-like blood vessels. Necrosis, calcification, and vascular extension may be present in the central area. Immuno-histochemical stains provide definitive diagnosis with the smooth muscle stains actin and vimentin being positive.
Prenatal detection with ultrasound is difficult with only three cases previously reported. Disruption to the sonographic appearance of organs, presence of abdominal mass and abnormal or limited fetal movements between 30 and 32 weeks’ gestation may point towards IM.
Tranexamic acid
Tranexamic acid (TXA) may be a useful agent to consider in emergency bleeding protocols in neonatal units. TXA is routinely used in children undergoing major cardiac and spinal surgeries and in cases of trauma. Evidence for its use in the neonatal population is limited.
TXA is a synthetic lysine analogue that inhibits conversion of plasminogen to plasmin by preventing plasminogen from binding to the fibrin molecule. TXA also inhibits plasmin activity directly. TXA inhibits fibrin cleavage, thus reducing the risk of haemorrhage. Metabolism of TXA in the liver is low, renal clearance amounts to 95% and the half-life is approximately 2.3 hours. There are case reports of seizures following high doses of TXA. Other common adverse effects include renal injury, renal failure and other neurological events. Reduction to the lowest required dose to prevent fibrinolysis should be used in patients with renal dysfunction.
Treatment regimens for use in neonates are based on extrapolated data from use in adults and children. As per the guidelines available for use in children and adults,
the suggested dose of TXA in neonates is a loading dose of 10-15mg/kg over 10 minutes, followed by infusion of 2mg/kg/hour for eight or more hours, or until bleeding ceases. Loading doses of 100mg/kg are given in cardiac surgeries. An intravenous preparation of TXA can also be used as a compress to control surface bleeding from haemangiomatous lesions, where compression/tourniquet fails to stop the bleeding.
Conclusion
This case reports the unusual clinical presentation of a congenital myofibroma in an immediate neonatal setting. The baby presented with a significantly bleeding lesion at birth and this adversely affected the outcome of the baby, despite aggressive resuscitation, optimal stabilisation and rapid transfer to a surgical centre for definitive management (ie excision of the lesion to stop major haemorrhage). Some mitigation strategies (using a tourniquet, clingfilm, etc) were used to stop the surface bleeding from the lesion, but in vain.
Neonatal medical emergencies like this case pose a conundrum to the practitioners forcing them to choose between limb or life. The learning point from this case is that neonatal units should have access to emergency bleeding protocols to deal with such major haemorrhages, similar to adults. There shall always be a need to involve the transfusion lab and paediatric haematologists in managing a major haemorrhage and the associated effects of disseminated intravascular coagulation. There is a role for the use of TXA as a last resort in cases of extensive bleeding unresponsive to conventional therapies.
This case also demonstrates the absolute importance of inter-hospital team working, including the regional specialist transport team and paediatric surgical team at the regional quaternary centre.
The use of telemedicine in supporting clinical management in such a setting cannot be overemphasised. Use of remote telehealth equipment could help in effec-tively supporting the clinical staff at the local hospital and establish a potent means of communication/support to the parents from the specialist teams based off-site.
Parental consent
Consent for publication was obtained from the parents of the patient.
Or read this article in our
Tablet/iPad edition
- Congenital myofibroma presenting at birth is a rare clinical finding.
- Ulcerating myofibromatous lesions can have significant consequences if not managed urgently, especially due to the risk of major bleeding and subsequent multi-organ failure.
- This case supports the need for devising major haemorrhage protocols in neonates.
Also published in Infant: