Neonatal herpes simplex virus infection: maternal risk stratification and management of at-risk neonates
Herpes simplex virus infection during the neonatal period is a relatively rare but serious diagnosis, associated with significant morbidity and mortality. Stratified management of mothers whose infants are at risk of perinatal infection and associated neonatal actions should be considered on a case-by-case basis. In this article, we discuss current guidance and challenges to clinical practice using a case-based approach to aid learning.
Monica Arend-Trujillo
ST4 Paediatric Trainee, Northern Deanery
monica.arend-trujillo@nhs.net
Lauren Dhugga
ST3 Paediatric Trainee, Northern Deanery
lauren.dhugga@nhs.net
Neonatal Intensive Care Unit, Royal Victoria Infirmary, Newcastle upon Tyne
Neonatal herpes simplex virus (HSV) infection is rare, with an estimated incidence of 10 cases per 100,000 live births worldwide.1 It is associated with significant morbidity and mortality with a mortality rate of up to 26%.2,3 Identification of and stratified management of mothers whose infants are at risk of perinatal infection is recommended in the UK, guided by a combination of maternal and obstetric considerations. Stratification of the risk of perinatal infection and associated neonatal/paediatric actions remains a clinical challenge that must be considered on a case-by-case basis.
A hypothetical case for learning
A female infant was delivered at 37 weeks’ gestation following induction of labour for concerns about fetal growth restriction. The infant’s mother had received antenatal steroids the day before delivery.
The infant was delivered vaginally three hours after rupture of membranes weighing 3.05kg. She was born in good condition without any need for resuscitation. She stayed with her mother, who planned to breastfeed. The infant was reviewed at one hour with signs of respiratory distress (tachypnoea, nasal flare, subcostal recession) with oxygen saturations of 92%. She was admitted to the neonatal intensive care unit for non-invasive respiratory support. She was supported with continuous positive airway pressure for 12 hours with supplemental oxygen maximum 30% and received intravenous (IV) benzylpenicillin and gentamicin after a blood culture was taken.
At six hours of age, a history of herpes came to light and local guidance was reviewed along with antenatal documen-tation. There was a history of primary maternal genital herpes infection four weeks prior to delivery, treated with oral aciclovir 400mg three times a day until delivery. There were no active maternal herpetic lesions at the time of delivery. According to this, the infant was categorised as high risk for perinatal transmission of HSV, due to primary infection less than six weeks before delivery (FIGURE 1). She was carefully examined and no skin, conjunctival or oral lesions were identified. She was commenced on empirical aciclovir 20mg/kg IV three times a day and investigations were sent as per the high-risk group (FIGURE 2). A full blood count, coagulation and liver function tests were requested and were normal. A blood sample was sent for HSV-1 and 2 polymerase chain reaction (PCR) and again 48 hours later; HSV was not detected in either sample. Swabs for HSV PCR were sent from the mouth, conjunctiva and skin at 48 hours and were negative. A lumbar puncture was performed at 36 hours of age and cerebro-spinal fluid (CSF) was sent for HSV-1 and 2 PCR. The results were negative.
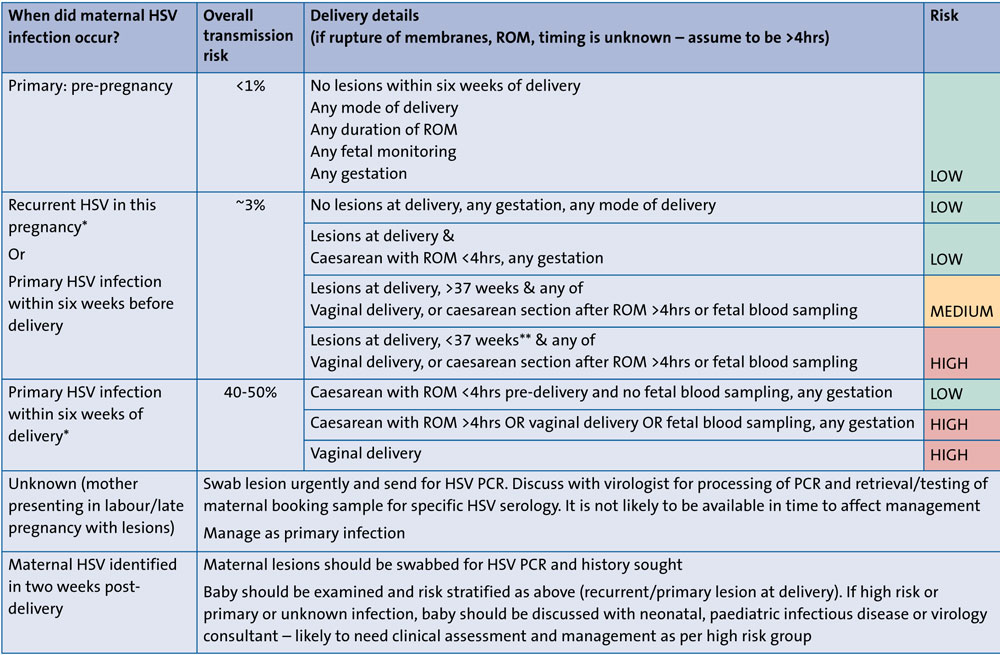
FIGURE 1 Local guidance on risk stratification according to timing of HSV infection in the mother and the labour and delivery details.10 *Although antenatal aciclovir suppressive therapy reduces maternal HSV shedding, it does not change risk stratification. **It is unknown at what gestation HSV Ig transfer is adequate for protection. Note that 28 weeks is the cut off used in UK guidance for protection from varicella-zoster virus infection.11
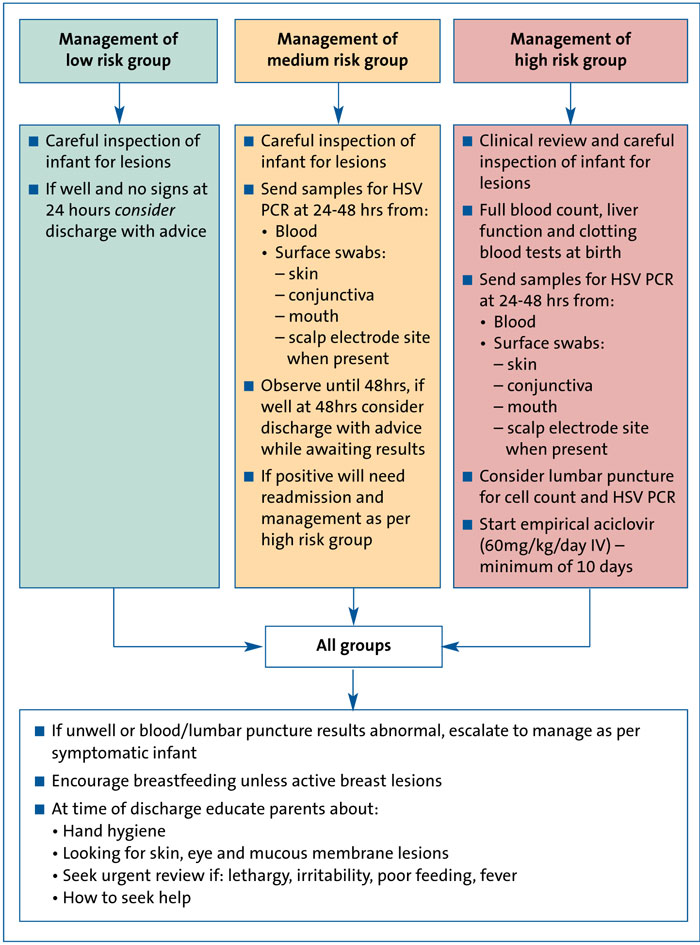
FIGURE 2 Management of at risk neonates according to risk stratification (adapted from Newcastle upon Tyne NHS Foundation Trust guidance).10
The infant completed a 10-day course of empirical IV aciclovir at 20mg/kg/dose three times a day. She was re-cannulated multiple times during this course. She remained well throughout admission with no skin manifestations or any other clinical concerns. She was cared for on the postnatal ward from day two of life and was discharged home on day 10 of life, with advice. The mother remained an inpatient with her on the postnatal ward and was upset by the unexpected, prolonged admission and multiple painful procedures her baby endured.
HSV-1 and HSV-2
HSV-1 and HSV-2 are alpha herpes viruses belonging to the family Herpesviridae, derived from the Greek word herpein (‘to creep’).4 They are enveloped viruses with a double-stranded DNA genome. The virus establishes latency after a primary infection, and periodically reactivates causing recurrent symptomatic disease. Importantly it can also cause asympto-matic viral shedding that is clinically unapparent.5 HSV-1 and HSV-2 can both cause genital infection, with HSV-1 being more classically associated with orolabial lesions and HSV-2 being the more common cause of genital lesions. More recently, however, HSV-1 has become the predominant virus causing genital herpes, responsible for up to 78% of genital herpes in some populations of young women.6 When considered alongside the decreasing HSV-1 seroprevalence in adolescents and young adults, these trends mean that an increasing number of young people are without protective HSV-1 antibodies at the time they become sexually active.7
Maternal genital HSV infection
When an individual with no prior HSV-1 or HSV-2 exposure acquires either virus in the genital tract, a first-episode primary infection occurs. After primary infection, viral reactivation from latency can result in a recurrent infection. These recurrences occur in the presence of pre-existing antibodies. Approximately two-thirds of women who acquire genital herpes during pregnancy have no symptoms to suggest a genital HSV infection. This is consistent with the finding that 60-80% of women who deliver an HSV-infected infant have no evidence of genital HSV infection at the time of delivery and have neither a past history of genital herpes nor a sexual partner reporting a history of genital herpes.8
Assessing risk of neonatal HSV infection
Perinatal risk factors alter the risk of neonatal acquisition of HSV. Some of these are modifiable and should be discussed with the mother antenatally where the risk is recognised (TABLE 1).9

TABLE 1 Risk factors for neonatal acquisition of HSV.9
Risk stratification according to maternal and perinatal factors
FIGURE 1 shows an example of a risk stratification model used at Newcastle upon Tyne NHS Foundation Trust. Infants are grouped as low, medium or high risk according to the timing of maternal infection, as well as labour and delivery details.
Management of at-risk asymptomatic neonates
FIGURE 2 demonstrates the management of at-risk neonates according to risk stratification, adapted from Newcastle upon Tyne NHS Foundation Trust guidance.10
Neonatal HSV infection
Neonatal infection with HSV is a potentially devastating complication of maternal genital herpes. It is rare but is associated with considerable morbidity and mortality. The majority (>85%) of neonatal herpes infections occur from exposure to HSV-1 or HSV-2 shed in the genital tract during delivery. Neonatal herpes infection due to a reactivated maternal infection is possible but the risk is low because of the presence of protective maternal IgG antibodies, which cross the placenta to afford immunity to the neonate. The risk of neonatal herpes infection is considerably greater for primary maternal infections close to term, as in our hypothetical case, when the virus is shed from the genital tract but maternal IgG antibodies have yet to be produced.1
Globally, the annual number of neonatal herpes cases during 2010-15 was estimated to be 14,257 of which approximately two-thirds (9,911 cases) were due to HSV-2, and a third (4,346 cases) were due to HSV-1.1 The global rate of neonatal herpes when averaged across all regions was estimated to be 10·3/100,000 live births.1 Neonatal herpes is rare in the UK, in contrast to some European countries and the USA. Active surveillance by the British Paediatric Surveillance Unit between 1986 and 1991 found 76 cases over the five-and-a-half-year surveillance period with an incidence of 1.65/100,000 live births annually.12 Subsequent surveillance from 2004 to 2006 showed an approximate doubling of incidence with 86 cases seen over the three-year surveillance period. This increase may reflect the rise in the prevalence of sexually transmitted infections, demographic and social changes within the general population and improvements in recognition of symptoms and diagnostic techniques.13 A more recent study in a single UK tertiary centre between 2006 and 2013 showed an incidence of neonatal infection at 17.5/100,000 live births, which is markedly higher than previous estimates in the UK.14
Mode of transmission of neonatal HSV
The majority of neonatal infections result from perinatal exposure during delivery (TABLE 2).15

TABLE 2 Types of HSV acquisition by the newborn.15
Disease classification and clinical manifestations of neonatal HSV
HSV infection acquired in the peripartum or postnatal period is classified by extent of disease, with disease classification being predictive of both mortality and morbidity. (TABLE 3). Clinical manifestations can be classified into three groups:
- Skin, eye, and/or mouth (SEM) disease
- Central nervous system (CNS) disease
- Disseminated disease.12
Treatment of neonates stratified at high risk for HSV infection
For infants deemed at high risk for HSV infection:
- Begin empirical IV aciclovir at a dose of 60mg/kg/day in three divided daily doses in high-risk exposures (FIGURE 1) or where there are clinical signs of neonatal HSV infection (TABLE 3). Continue IV aciclovir for a minimum of 10 days (as in the hypothetical case described).
- If the result of a diagnostic evaluation is positive, continue IV aciclovir for 14 days in SEM disease or at least 21 days in CNS disease or disseminated disease.
- In CNS disease (positive lumbar puncture or clinical signs without successful lumbar puncture), repeat HSV PCR on CSF near the end of the 21-day course of treatment to ensure viral clearance.
- After completion of parenteral therapy for CNS, disseminated, or SEM disease, administer a suppressive course of oral aciclovir at a dose of 300mg/m2/dose three times a day for six months.
- Monitor absolute neutrophil count at the second and fourth week of suppressive therapy and then monthly throughout the remainder of the treatment period.7
- If a diagnostic evaluation does not isolate HSV and the infant remains clinically well, complete 10 days of IV aciclovir. At the time of discharge give specific advice about signs of neonatal HSV infection.
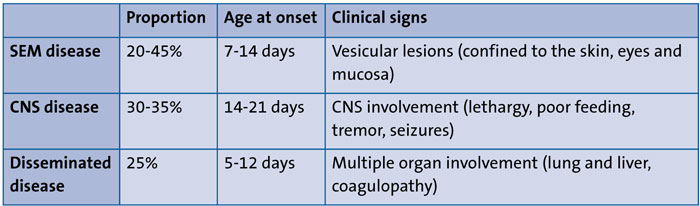
TABLE 3 Clinical manifestations of neonatal HSV.16
Prognosis
The introduction of aciclovir as an effective non-toxic antiviral treatment in the 1980s has improved the outcome of neonatal HSV and management of high-risk cases awaiting results. PCR testing allowing rapid and accurate diagnosis, has also helped.8 Mortality remains highest in the disseminated group, though this has improved from 85% to 29%.8 In the CNS group, mortality has improved from 50% to 4%. Overall neonatal HSV mortality over a 10-year-period in the USA was 7.9%.2 Morbidity has improved too, with 83% of infants with neonatal disseminated HSV developing normally at one year of age, compared with 50% previously.
Although neonatal HSV is a treatable disease, recognition and initiation of treatment can sometimes take time.17 As with many neonatal illnesses, infants can present with vague, non-specific symptoms and, as described, there is often no history of symptomatic HSV infection in the mother.18 HSV infection is sometimes considered in neonates with bacterial culture-negative suspected sepsis or those who deteriorate despite broad-spectrum antibacterial therapy, by which point dissemination may have occurred.
Neonatal HSV infection is rare but potentially devastating. Where there is an identified risk of perinatal HSV infection there are many aspects to consider for managing pregnancy, labour and postnatal care of both the mother and infant. This discussion should be initiated by the obstetric team as soon as the risk is identified to allow time to plan for antenatal antiviral therapy and the most appropriate method of delivery. The possibility of a prolonged postnatal hospital stay should also be discussed. The psychosocial impact of a new diagnosis of HSV is recognised and may itself require counselling.19 Advising a caesarean section where there is a primary infection in the third trimester is the gold standard and should be encouraged, though maternal wishes must be considered.
Maternal antiviral medication should be prescribed to treat primary infection in pregnancy. Where primary infection is in the third trimester and the baby is born vaginally, empirical aciclovir should be started for the infant while a workup to look for HSV including blood and surface swabs for HSV PCR are awaited.
A lumbar puncture should be performed in an unwell neonate where HSV infection is suspected and should be considered in any infant at high risk of perinatal HSV acquisition, except when clinically contraindicated.
The duration of aciclovir in the well but at-risk infant is typically 10 days, although there is little evidence to support this duration without any isolation of HSV on surface swabs or serum PCR. More research into the optimum duration of antiviral treatment in this population is needed before current practice can be reviewed.
Acknowledgement
The authors are grateful to Dr Janet Berrington, Consultant Neonatologist and Dr Sheila Waugh, Consultant Virologist at the Royal Victoria Infirmary, Newcastle upon Tyne Hospitals NHS Foundation Trust, for their specialist input and manuscript review.
Or read this article in our
Tablet/iPad edition
- Antenatal detection of HSV must be communicated across obstetric and neonatal teams for optimal care for mother and neonate.
- Identifying and managing risk factors antenatally, including empirical maternal antiviral treatment, can reduce vertical transmission of HSV.
- Risk stratification should be used to guide investigations and indication for empirical antiviral treatment in neonates.
- Women with HSV infection in pregnancy should be counselled about expected management of the neonate and potential prolonged hospital stay.
Also published in Infant: