Management challenges in Shwachman-Diamond syndrome
Shwachman-Diamond syndrome (SDS) is a rare disorder, characterised by bone marrow dysfunction, exocrine pancreatic insufficiency, failure to thrive and skeletal abnormalities. It is most commonly diagnosed in early childhood after the development of malabsorption and neutropenia with associated risk of infection. SDS is mainly inherited in an autosomal recessive manner, with most pathogenic variants found in the SBDS gene. This case report describes the challenging clinical presentation of a neonate born at 35 weeks’ gestation with SDS and homozygous variants in the EFL1 gene.
Mohamed Omer1
Paediatric Registrar
Muna Ewadh2
Obstetrics and Gynaecology Registrar
Melody Grace Redman3
Clinical Genetics Registrar
Uma Rajesh2
Obstetric Consultant
Nicola Mullins4
1Neonatal Unit, Hull University Teaching Hospitals NHS Trust
Neonatal Consultant
2Department of Obstetrics and Gynaecology, Hull University Teaching Hospitals NHS Trust
3Yorkshire Clinical Genetics Service, Leeds Teaching Hospitals NHS Trust
4Neonatal Unit, Leeds General Infirmary, Leeds Teaching Hospitals NHS Trust
Antenatal management
A24-year-old woman, primigravida in a consanguineous marriage, booked at eight weeks’ gestation. At booking, blood tests revealed low levels of pregnancy associated plasma protein-A (PAPP-A; 0.30MoM). A routine anatomy scan at 20 weeks identified short long bones, a small cerebellum, an echogenic bowel, an atrioventricular septal defect and severe intrauterine growth restriction (IUGR; <3rd centile). The kidneys, ureters and bladder appeared normal. Invasive testing was initially declined by the parents.
A further growth scan confirmed IUGR (femur length <3rd centile) and severe oligohydramnios. Doppler assessment of the umbilical artery, ductus venosus and middle cerebral artery (MCA) were normal. There were no concerns regarding anaemia in the antenatal period at any stage with normal MCA peak systolic velocity throughout (which correlates excellently with fetal anaemia).1
Amniocentesis was undertaken at the couple’s request at 25 weeks’ gestation, which highlighted a normal quantitative fluorescent polymerase chain reaction (QF-PCR) and array comparative genomic hybridisation (CGH). The risks of a rare genetic syndrome underlying the antenatal findings were explained to the parents. The genetics and fetal cardiology team were involved. The couple were committed to the pregnancy and were supported extensively via the fetal medicine team.
Birth
At 35+5 weeks’ gestation an emergency lower segment caesarian section was performed following pathological cardio-tocography with antepartum haemorrhage. At birth the male baby weighed 1,310g (<0.4th centile; -3 standard deviations), and occipitofrontal circumference measured 31cm (50th centile). He was noted to have joint contractures, bilateral talipes, micropenis and cryptorchidism. The placental weight was 231g (<3rd centile) with hyper-coiling of vessels.
Neonatal management
Following intubation and surfactant administration at birth, this baby remained ventilator-dependent (conven-tional and high frequency with inhaled nitric oxide) due to a combination of lung hypoplasia and persistent pulmonary hypertension. He also required full inotropic support. He developed progressive bone marrow failure,2 requiring multiple blood product trans-fusions, neutropenia and subsequently leukopenia. He was managed for febrile neutropenic sepsis. Enterobacter cloacae was identified in the blood. Methicillin-resistant Staphylococcus aureus (MRSA) and Stenotrophomonas were identified in the sputum. TORCH screening, B12/folate levels and parvovirus PCR were unremarkable.
Postnatal echocardiography showed multiple small ventricular septal defects, severe tricuspid regurgitation, mild mitral regurgitation and pulmonary hypertension. The baby was commenced on sildenafil.
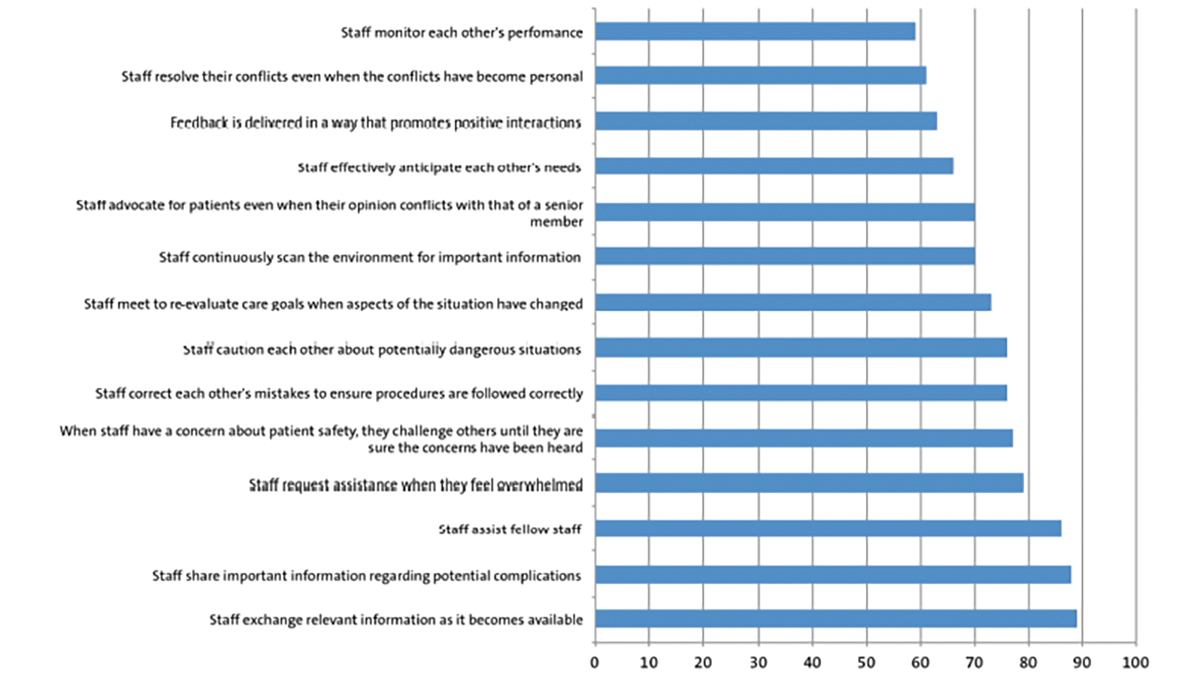
He developed abdominal distension and feeding intolerance, with no surgical cause found. He remained dependent on total parenteral nutrition. His 17-hydroxyl progesterone level was normal. He had a cranial ultrasound and ophthalmological evaluation, which were normal. A skeletal survey demonstrated significant osteopenia, short long bones, a bell-shaped thorax, a poorly ossified large skull vault and abnormal broadening ribs with cupping (FIGURE 1). The respiratory, haematology, genetics and renal teams were involved in joint management.
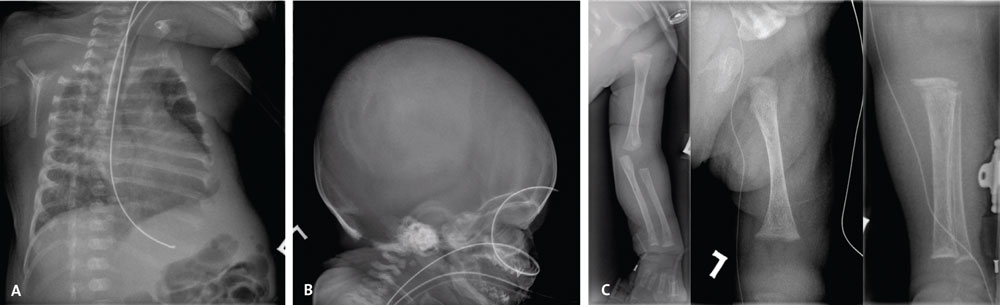
FIGURE 1 Skeletal survey findings. (A) Chest X-ray: showing abnormal ribs broadened anteriorly with cupping. (B) Skull X-ray: the skull vault is poorly ossified. (C) Limb X-rays showing shortened long bones, which are osteopenic and have irregular metaphyses.
His bone marrow suppression was initially considered related to the severe IUGR, placental insufficiency and bone marrow failure. A bone marrow aspirate showed aplastic marrow with low level haemophagocytosis, reduced cellularity, and undetermined bone marrow aplasia with no blast cells.
Diagnosis
Obtaining a sample for genetic testing≤sup>3proved complex as the baby received multiple blood product transfusions. Ultimately, a skin biopsy was required. Rapid trio exome sequencing, using DNA samples from the baby and both parents, revealed a molecular diagnosis. The baby was homozygous for a likely pathogenic EFL1 missense variant (NM_024580.5[EFL1]:c.2909G>A [p.Arg970His]), consistent with a diagnosis of SDS type 2.4,5 Both parents carried one copy of the same variant; the baby inherited one variant from each parent.
The baby’s condition acutely deteriorated on day 48 of life with severe sepsis (Stenotrophomonas maltophilia gram negative sepsis). He was commenced on granulocyte-colony stimulating factor (G-CSF) but sadly passed away soon after the diagnosis of SDS had been made.
Discussion
SDS is usually inherited in an autosomal recessive manner, with pathogenic variants most commonly in the SBDS gene.6,7 Pathogenic variants in EFL1 are rarely the cause, and account for <1% of SDS cases.7 Very rarely, SDS can be autosomal dominant, for example with a de novo pathogenic variant in SRP54.8 In this case, the EFL1 variant inherited from each parent was identical, due to the history of consanguinity.
The parents were counselled about a 25% recurrence risk for each future child to be affected with SDS. Options such as pre-implantation genetic diagnosis or antenatal testing through chorionic villus sampling were offered. Close surveillance of future pregnancies via fetal medicine was also offered.
At the time of writing, there are at least 461 different genetic skeletal disorders, with 437 disease-causing genes identified.9 These disorders are classified into 42 different groups; SDS falls within the ‘metaphyseal dysplasia’ group.9 A proportion of these conditions may present with signs during the antenatal period. This presentation further emphasises the importance of collaboration between the fetal medicine and clinical genetics departments during the antenatal period when skeletal abnormalities are identified during antenatal scans. Couples may not wish to undergo genetic testing during pregnancy for many reasons, but the clinical genetics team can still be involved. In some cases, it may be appropriate to seek advanced parental consent for storage of DNA from a cord sample. Postnatally, collaboration between neonatology, clinical genetics, and other relevant teams is key. This approach allows phenotyping of the baby, counselling of the parents, consideration of differential diagnoses, and appropriate testing to be offered.
Lessons learnt
The lessons learnt from this care are:
- Always store DNA prior to blood transfusions in complex settings.
- Normal antenatal genetic testing does not exclude a molecular diagnosis.
- Early multidisciplinary involvement from fetal medicine, obstetrics, genetics, radiology, haematology and neonatology are essential to obtaining a diagnosis enabling, for example, pre-implantation genetic diagnosis.
- Improved communication between genetics, fetal medicine and neonatology is required.
- Low platelets are not always directly related to IUGR.
- Consider SDS in neonates with pancy-topenia and possible skeletal dysplasia.
Parental consent
Written consent for publication was provided by the patient’s parent.
Or read this article in our
Tablet/iPad edition
- Standard antenatal genetic testing does not exclude a molecular diagnosis of SDS.
- Consider SDS in neonates with pancyto-penia and possible skeletal dysplasia.
Also published in Infant: