Do we know how to treat PDA with paracetamol? Current evidence on the pharmacokinetics and pharmacodynamics of paracetamol for hsPDA closure in extreme preterm infants
Paracetamol is increasingly used for the treatment of a haemodynamically significant patent ductus arteriosus (hsPDA) and is touted as a better alternative to ibuprofen, the current drug of choice in the UK. This review examines the evidence from randomised controlled trials (RCT) comparing paracetamol with ibuprofen, in terms of dose and pharmacokinetic and pharmacodynamic profiles. It appears that there is a lot to ascertain before paracetamol can be confidently used over ibuprofen for management of hsPDA in preterm infants.
Anupam GuptaConsultant Neonatologist1
anupam.gupta@mft.nhs.uk
Arin Mukherjee
Consultant Neonatologist1 and Honorary Senior Lecturer2
arindam.mukherjee@mft.nhs.uk
1Department of Neonatology,
St Mary’s Hospital, Manchester University NHS Foundation Trust
2University of Manchester
Background
Prolonged exposure to a hsPDA in extremely premature infants (<28 weeks’ gestation) can lead to significant mortality and morbidity. Ibuprofen remains the first-line treatment but paracetamol is emerging as a strong alternative.
The spontaneous closure rate of PDA is inversely proportional to gestation at birth. Infants born at <28 weeks’ gestation have the lowest chance of spontaneous closure of the PDA. The literature observes spontaneous PDA closure rates in only 34% of infants <28 weeks’ gestation.1 It is known that prolonged exposure to hsPDA for greater than 7-13 days, increases the incidence of bronchopulmonary dysplasia (BPD) or death.2 It is well established that the effects of non-steroidal anti-inflammatory drugs (NSAID, eg ibuprofen or indomethacin), in ductal constriction, decreases considerably beyond two weeks of age.2-4 It is postulated that non-prostaglandin pathway mech-anisms are involved in keeping ductal patency beyond this age.4 Most published trials showed the benefits of potential treatment when initiated within the first two weeks of life.2-5
Intravenous (IV) ibuprofen is the only licenced medication for management of PDA in the UK.6 Although there is an increased awareness of the benefits of paracetamol in PDA treatment, the exact mechanism by which paracetamol closes a PDA is not fully understood. It appears that paracetamol inhibits prostaglandin synthetase activity by acting at the peroxidase segment of the enzyme, as opposed to the cyclooxygenase segment.1
A recent national survey7 of UK neonatal intensive care units (NICU) carried out by the authors observed that 82% of NICUs in the UK use paracetamol to treat PDA. While the majority of units (65%) used it as a second-line treatment for PDA, 10% used it as a first-line treatment in preference to ibuprofen.
Available evidence from the literature suggests that the dose of paracetamol used to treat PDA varies between 30mg/kg/day and 60mg/kg/day, as does the duration – from three days to seven days. A recent systematic review8 of paracetamol recognised it as a promising alternative to ibuprofen, but was unable to provide practice recommendations due to the heterogeneity in the methodology and designs of the available clinical trials.
Aims and objectives
The objective of this review was to examine evidence on the dose, pharmacokinetic and pharmacodynamic profile of paracetamol and establish its current position regarding use for management of hsPDA in preterm infants.
We carried out this targeted literature review to improve understanding of the current evidence on the pharmacokinetic (with particular focus on therapeutic drug levels) and pharmacodynamic (efficacy, safety profile and drug regimens) properties of paracetamol in PDA management. It is envisaged that this will help neonatologists in their understanding of the clinical use of paracetamol and identify gaps in knowledge to encourage better designed clinical trials for the future.
Methodology
A thorough literature search of online databases was performed. The articles classed as RCTs on use of paracetamol for management of PDA were evaluated. We searched the online databases Medline, Embase and Cochrane using the keywords patent ductus arteriosus AND paracetamol. We then reviewed the results for articles on case reports, observational studies and RCTs. The exercise returned 46 articles on paracetamol for PDA management, which were mostly case series and case reviews. Twenty RCTs were identified; eight of these RCTs used an IV preparation of paracetamol and 12 RCTs used oral paracetamol. These RCTs were evaluated in our review.
Results
The dose, duration, route of administration of paracetamol for treatment of PDA remains variable across RCTs. A loading dose followed by a lower daily cumulative dose of IV paracetamol appears to have a better safety profile, while retaining similar efficacy in comparison to higher daily cumulative doses. Heterogeneity of the clinical trials and their trial designs preclude the use of therapeutic drug monitoring as a marker of safety and effectiveness for paracetamol treatment across all gestations of prematurity. The outcome and safety profile of paracetamol in the longer term remains unascertained.
Pharmacokinetics
While the half-life of paracetamol in adults is suggested to be 5.6 hours, pharmacokinetic studies in preterm and term newborn infants found these to be 4.6 hours and 2.9 hours respectively.9 In adults, paracetamol metabolism takes place in the liver by following pathways leading to non-toxic metabolites that are eventually eliminated by the kidneys:10
- glucuronidation (45-55%)
- sulphation (sulphate conjugation) (20-30%)
- N-hydroxylation and dehydration, then glutathione conjugation (<15%).
However, in extremely preterm infants the main mode of metabolism is sulphation11 and the glucuronidation metabolism of paracetamol increases with advancing gestational age.12 As the extent of glucuronidation and sulphation is dependent on the gestation and maturity of infants, a uniform standard serum level across all gestations becomes hypothetical. Hence, serum paracetamol levels remain an unpredictable marker to guide direct treatment.13 In addition, appropriate clinical trial designs and assays incorporating robust evaluation of gestation-specific serial serum concentrations, to determine optimal dose response threshold and toxicity, are yet to happen in extreme preterm infants.3,14
Allegaert et al11 carried out one of the most comprehensive works in producing pharmacokinetic evidence on IV para-cetamol use in neonates. The authors demonstrated that an increased volume of distribution supports the use of a loading dose in neonates. They also reported that size (described by patient weight) is the major co-variate contributing to paracetamol clearance variance in neonates as paracetamol clearance (mg/kg/hour) increases marginally throughout the neonatal life. The authors suggested a loading dose of 20mg/kg followed by 10mg/kg every six hours within the age range evaluated (32-44 weeks’ post-menstrual age) to achieve the desired mean concentration of the drug.
In vivo, the serum concentration of a drug at any given time is in a state of continuous dynamic equilibrium with its absorption, distribution, storage in tissues, liberation and elimination. Hence, any evaluation of the serum therapeutic concentration requires comprehensive pharmacokinetic compartment modelling. Compartment models in pharmacokinetic studies simulate these dynamic interplays of drug absorption, distribution and elimination. A single compartment linear disposition pharmacokinetic model (zero-order input, first-order elimination) is supposedly the least accurate evaluation, as it assumes a homogenous distribution of the drug in the body. The pharmacokinetic evaluation by Allegaert et al is based on a two-compartment model, which is better than a single compartment model.
In addition, the presence of factors such as renal disease, liver impairment and hyperbilirubinaemia, can all affect paracetamol metabolism and clearance. Any infant on enzyme-inducing anti-epileptic drugs (eg phenobarbitone, rifampicin, phenytoin) should be monitored closely with caution due to potential interaction with paracetamol.
To date, the majority of clinical trials on the use of paracetamol have relied on trial-and-error methods of drug dosage and serum levels rather than predicting the kinetic behaviour of paracetamol through comprehensive pharmacokinetic modelling.
In a very recent study by Cakir et al, the authors compared continuous infusion vs standard intermittent bolus infusion of paracetamol to treat PDA.15 In spite of obvious theoretical advantages of continuous infusion in achieving consistent therapeutic serum concen-trations, the study did not find any merit in the use of paracetamol infusion in comparison to intermittent bolus doses for the pharmacological treatment of PDA. Lower rates of PDA-related morbidity (BPD, necrotising enterocolitis, NEC, and need for ligation) were noted with intermittent dosing.
Pharmacodynamics
Unlike NSAIDs, which have a vaso-constrictive mechanism of action, paracetamol exerts a vasodilatory effect.13 This observation may explain the fewer side effects encountered with paracetamol use. Vasoconstriction is thought to be the primary causative factor for renal impairment, cerebral ischaemia and NEC seen with some NSAIDs.
While vasodilation raises concerns of clinical hypotension following paracetamol administration, it has not been a major issue in the neonatal population.12 It is postulated that the metabolite responsible for the vasodilatory effects is NAPQI (N-acetyl-p-benzoquinone imine). As less than 15% of paracetamol is metabolised via this route and NAPQI is detoxified rapidly by glutathione in the liver and excreted in the urine, under physiological conditions its impact is minimal and short-acting.13
Route of administration
A recent UK national survey noted that 73% of NICUs use only an IV preparation of paracetamol and 26% of units use either an IV or enteral preparation, depending on the fed status of their infants.7
It also appears that oral paracetamol has similar efficacy to oral ibuprofen.10,16 However, the oral route probably does not represent the optimal choice for extremely low birth weight infants. Gut immaturity, poor enterohepatic circulation together with feed intolerance in these infants is likely to contribute to reduced efficacy and unpredictable paracetamol toxicity.
A recent systematic literature review tentatively suggests that, while oral paracetamol appears to be effective in infants weighing 1,501g to 2,500g, more elaborate and comprehensive RCTs are needed to ascertain its effects in more premature infants.8
Dose
Following the first report in 2013 by Oncel et al,17 that an IV dose of paracetamol at 15mg/kg every six hours (daily cumulative dose of 60mg/kg/day) can be used in extremely premature infants successfully to close a PDA, many observational studies and some RCTs have used it with success. These trials neither clarified on the optimal dosage regimen nor did they come up with the timing of the initiation of therapy related to paracetamol use in neonatal age groups.18 There is no consensus on the dose, duration and the route of admini-stration of paracetamol in hsPDA. Hence, irrespective of the route of administration, its off-label use is also affected by widely variable dosages and duration (between three and seven days).
Tekgunduz et al19 reported in a retro-spective study that they encountered raised transaminase enzyme levels with IV administration of paracetamol at doses of 15mg/kg six hourly (cumulative dose 60mg/kg/day). The authors reduced the dose to 10mg/kg every eight hours and reported the closure rate of PDA with this lower dose at 83.3%.
Terrin et al20 recommended a limitation of dosing to 45mg/kg/day to avoid potential toxicity and still had similar efficacy rates.
Enteral
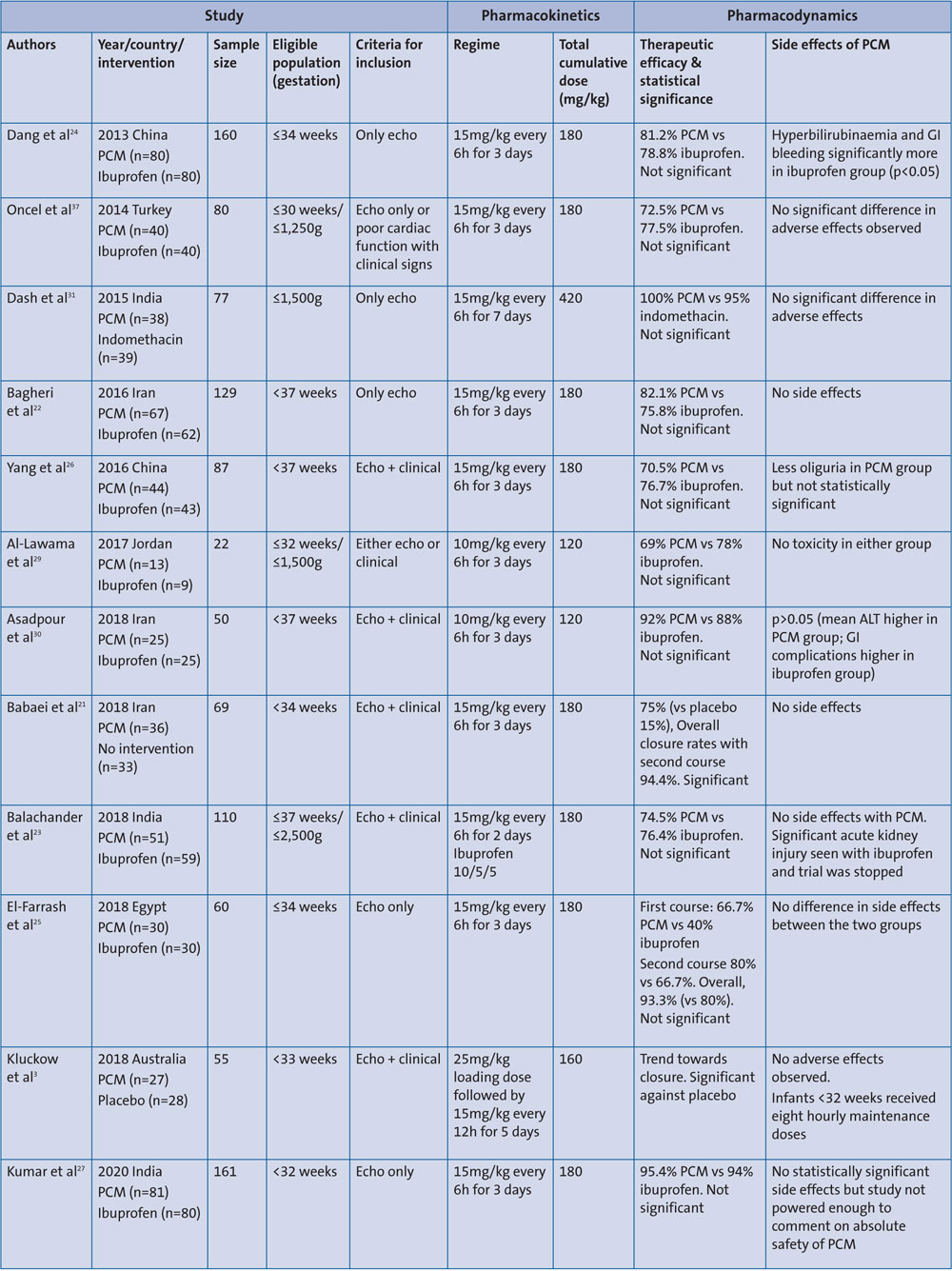
We identified 12 RCTs on the use of oral paracetamol for PDA treatment (TABLE 1). Oral paracetamol appears to be well toler-ated and there were no significant side effects in comparison to placebo. Efficacy was noted to be comparable to ibuprofen. The majority of the researchers used a dose of 15mg/kg every six hours for three days21-28 giving a cumulative dose of 180mg/kg over the course of three; the others opted for a dose of 10mg/kg every six hours for three days.29,30 Kluckow et al3 and Dash et al31 went further to give the course for five and seven days. A review of the studies can conclude that the dose and the duration of the oral treatment in the studies so far remains widely variable.
Of note, the median gestational age for RCTs using IV paracetamol was 27.6 weeks, whereas the median gestational age used in all studies for oral paracetamol was 31.1 weeks.
TABLE 1 A summary of the pharmacokinetics and pharmacodynamics from RCTs using oral paracetamol for PDA management. Key: PCM=paracetamol; echo=echocardiography; GI=gastrointestinal; ALT=alanine aminotransferase.
IV administration
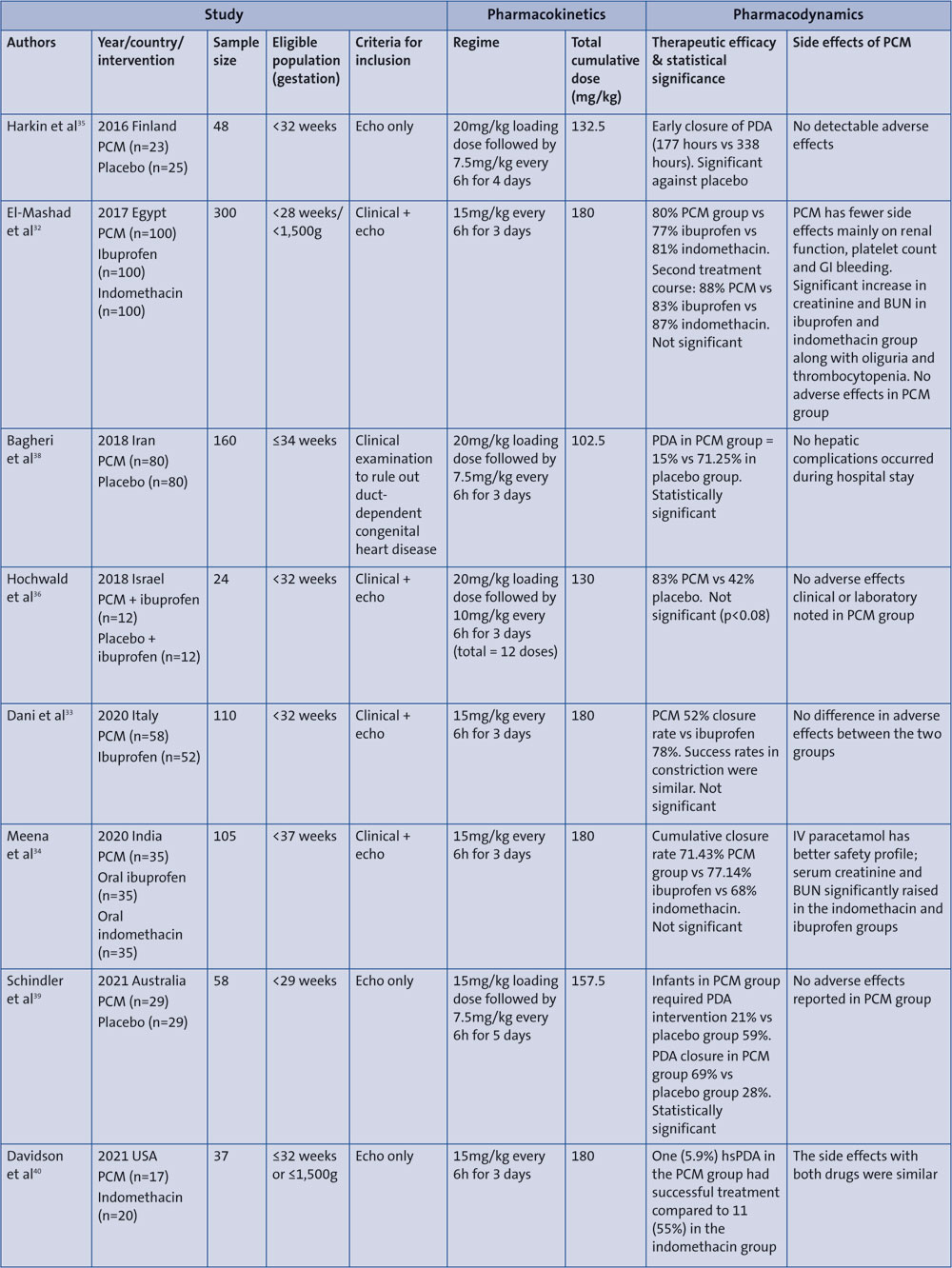
There are eight RCTs that investigated the use of paracetamol for hsPDA closure (TABLE 2). El-Mashad, Dani and Meena et al32-34 used doses of 15mg/kg every six hours for three days, which translates into a cumulative dose of 180mg/kg for the full course of paracetamol. Harkin and Hochwald et al35,36 opted to use a loading dose of 20mg/kg followed by 7.5mg/kg every six hours for four days and 10mg/kg every six hours for three days, leading to similar cumulative doses of 130-133.5mg/kg.
TABLE 2 Summary of pharmacokinetics and pharmacodynamics from RCTs using IV paracetamol for PDA management. Key: PCM=paracetamol; GI=gastrointestinal; BUN=blood urea nitrogen; echo=echocardiography; hsPDA=haemodynamically significant patent ductus arteriosus.
Toxicity and side effect profile
As paracetamol can induce hepatocellular necrosis, perhaps the best early marker to determine hepatocellular injury is through estimation of liver transaminases (such as aspartate transaminase, AST, and alanine aminotransferase, ALT). Tekgunduz et al reported significant increase in transaminases above the pre-treatment levels (AST and ALT rose to 260 and 180U/L from pre-treatment levels of 43 and 11, respectively).19 This was observed at an IV paracetamol dose of 15mg/kg every six hours and reversed to normal levels over five days once treatment was discontinued. However, it appears that neonates tend to suffer less from the hepatotoxic effects of paracetamol than do older children.41
Overt liver failure is extremely rare too. We found only one case report of acute liver failure in a term neonate receiving oral paracetamol at 10mg/kg/dose every four hours for three days (daily dose 60mg/kg; cumulative dose 180mg/kg).42 The infant responded well to IV
N-acetylcysteine. While this case report highlights the risk of severe hepatotoxicity in neonates after multiple doses of paracetamol for more than 2-3 days, such incidences have remained exceedingly rare.
Apart from this, we did not find any other case reports of term or preterm infants suffering from major side effects during treatment with paracetamol. The RCTs conducted to date depict an overall very safe profile for paracetamol in PDA treatment. None of the 20 RCTs using either IV or oral paracetamol administered in a total of 886 preterm infants, reported any serious untoward incidents or reactions (TABLES 1 and 2).
Long-term safety profile
Bauer et al raised concerns about the possibility of adverse long-term neurological outcome in their ecological study by demonstrating an association between pre- and perinatal use of paracetamol and autistic spectrum disorders.43 In 2017, a Swedish study by Bornehag et al reported a possible association of maternal pre-exposure to paracetamol and language delay in a 30-month-old girl.14 On the other hand, Oncel et al reported no significant difference in neurological outcome among preterm infants who received either paracetamol or ibuprofen at 18-24 months corrected gestational age.28 Similarly, Harma et al found better cerebral oxygenation and neuroprotective effects with postnatal paracetamol use in extremely premature infants with PDA.44
Drug levels
While there is sufficient evidence to support that serum paracetamol levels are a reliable marker of paracetamol toxicity in children and adults, their role in clinical management of preterm infants remains very limited, as far as their relationship to efficacy or toxicity is concerned. McPherson et al45 analysed paracetamol serum concentrations in preterm infants treated intravenously for PDA and found no correlation between paracetamol serum levels with infant demographics, hepatic transaminases during treatment or duct size at treatment completion.
This can be partly explained by our current understanding of pharmaco-kinetics that the metabolism of paracetamol is dependent on the level of prematurity and hence on gestation.
As there is no established or validated nomogram on gestation-specific serum paracetamol levels, any attempt in interpreting serum drug levels across the board will be of limited value.
The difficulty in interpretation also extends to the type of serum levels (eg peak, trough or steady state levels). The serum levels used in the literature for extremely premature infants are quite variable in relation to administration of the dose and timings. Therefore, future studies should not only be designed to evaluate gestation-specific nomograms in extremely premature infants but should also work on improving the understanding of clinically relevant timing of levels.
Limitations
Although the RCTs carried out so far have helped to improve our understanding, it is still not any easier for clinicians to make evidence-based decisions for treating a PDA with paracetamol. This is partly attributed to the heterogeneity of the studies. Several aspects of paracetamol use in the neonatal age group remain unclarified in these trials. In particular:
- optimal dosing regimen
- time of initiation of therapy
- route of administration
- direct comparison of different dosing regimens.
The RCTs also have significant limitations, such as:
- uniformity in defining the determinants of hsPDA
- echocardiographic parameters for hsPDA evaluation
- variable inclusion criteria
- inadequate sample power calculations
- absence of intention-to-treat analyses.
While our review has covered case reports, case series and observational studies to identify safety issues, we only used RCTs when evaluating the variability in the dosage regimen and monitoring of safety and effectiveness of paracetamol.
Summary
Paracetamol is establishing itself as an alternative to the conventional treatment of hsPDA by NSAIDs. Although several RCTs have evaluated the efficacy and safety of ibuprofen and paracetamol, a consensus on the practice recommendations has been difficult to establish given the heterogeneity of these trials. The two national surveys conducted in the UK continue to highlight the variability in dosage, duration of treatment and monitoring of infants during paracetamol treatment for PDA closure.7,46
The dose, route of administration, efficacy reports and short- and long-term implications still remain highly variable.6 The majority of RCTs involving oral dosing of paracetamol to treat hsPDA have used 15mg/kg/dose (cumulative dose of 60mg/kg/day) safely and effectively. However, it appears that lower cumulative doses of IV paracetamol (40-50mg/kg/day) may be equally efficacious to higher daily cumulative doses (60-70mg/kg/day) while assuring a better safety profile, given the hepatic and renal immaturity in preterm infants. The data from RCTs combined with experience of its use in older preterm infants, consolidates our understanding of its excellent safety profile.
The review, however, casts doubt on the reliability of therapeutic drug level monitoring of paracetamol as a marker of its safety or efficacy. Data on gestation-specific dosing nomograms should be sought through studies to describe its time concentration profile.
Finally, well-designed multicentre RCTs allowing exploration of the predictive kinetic behaviour of paracetamol through pharmacokinetic multi-compartment modelling are necessary. This will provide the much-needed evidence to help neonatologists confidently use paracetamol as a first-line medication to treat hsPDA.
Conclusion
Paracetamol remains a potential second-line alternative to NSAIDs in managing hsPDA. However, assurance regarding its optimum dosage, route of administration, short- and long-term implications from studies conducted so far is limited.
Contributions
AM (ORCID iD: 0000-0002-3933-7028) and AG conceived and designed the project, reviewed and analysed the data and co-wrote the article.
Or read this article in our
Tablet/iPad edition
- Lower cumulative doses of IV paracetamol may be equally efficacious to higher daily cumulative doses while assuring a better safety profile.
- Our review casts doubt on the therapeutic drug concentration of paracetamol as a marker of its safety or efficacy.
- Well-designed multicentre RCTs allowing exploration of predictive kinetic behaviour of paracetamol are needed to establish its safety and efficacy over NSAIDs.
Also published in Infant: