Pseudotumoral cystitis: a case of acute urinary retention in a newborn
Pseudotumoral cystitis is a subtype of eosinophilic cystitis – a rare inflammatory bladder condition with a wide range of clinical presentations. In this article, we describe the case of a male infant with an antenatally diagnosed unilateral hydronephrosis, presenting at 24 hours of age with urinary retention. An initial urinary tract ultrasound scan showed a hyperechoic bulge at the bladder base with persistent hydronephrosis. As far as we can tell, this is the second case of neonatal pseudotumoral cystitis and the ninth case of paediatric pseudotumoral cystitis to be reported in the literature.
Arameh Aghababaie
Paediatric ST2 Doctor
a.aghababaie@nhs.net
Hesham El Sayed
Consultant Neonatologist
Neonatal Unit, Newham University Hospital
(Barts Health NHS Trust), London
Background
When a baby is born, midwives and neonatal doctors alike repeatedly give the same safety-netting advice to parents:
- feed the baby every 2-4 hours
- make sure they pass urine within 24 hours of birth
- make sure they pass meconium within 48 hours of birth.
There is a reason why we provide this advice; the rare case that a baby cannot feed, or pass urine or meconium can indicate significant pathology. So, what do you do when you actually have been informed that a baby has not passed urine within 24 hours of birth? In this article, we describe such a case and how we investigated and managed the baby until the very rare cause of the baby’s urinary outflow obstruction came to light.
The case
A male infant was born to a 38-year-old Indian female at 39+2 weeks’ gestation via spontaneous vaginal delivery, weighing 3,300g (29th centile). The mother’s labour was induced due to polyhydramnios. The only other scan abnormality of note was a right renal pelvic dilatation (15.1mm) and a mildly dilated right ureter and mild bladder wall thickening with a ‘keyhole sign’ (dilated proximal ureter and thickened bladder wall resembling a keyhole) at the 35-week scan. The remainder of the scan and the Doppler assessments were normal.
The mother’s blood group was A+ with no atypical antibodies, her serology and virology were all negative (ie protective for the baby), and her antenatal TORCH (toxoplasmosis, rubella, cytomegalovirus, herpes simplex, and human immuno-deficiency virus) screen was negative.
The mother did not have any medical problems during pregnancy and she did not take any medications apart from folic acid and prenatal vitamins. The mother was gravida 4 and para 3; her previous children were all well. The parents were non-consanguineous and there was no relevant family history.
The baby was born in good condition with Apgar scores of 9, 10, 10 at one, five and 10 minutes and he did not require any resuscitation. There were no risk factors for early onset neonatal sepsis. In light of the right renal pelvic dilatation in the most recent antenatal scan, the baby was commenced on prophylactic trimetho-prim. The baby was transferred to the postnatal ward with the mother and an outpatient ultrasound scan of the kidneys, ureter and bladder (USS KUB) was organised for 7-10 days’ time, alongside an outpatient neonatal consultant follow-up. The parents were safety netted to seek urgent medical attention if the baby did not pass urine within 24 hours.
At 18 hours of age the mother and midwife were confident that the baby had not had any wet nappies since birth and therefore the neonatal team was informed. The baby was feeding well and had passed meconium since birth. Therefore, the mother was encouraged to continue breastfeeding and advised to massage the baby’s lower abdomen gently and to place wet cotton wool on the lower abdomen to stimulate the passage of urine.
At 24 hours of age, the neonatal team returned to conduct another review of the baby as he had still not passed urine. On examination, his airway was patent and the chest was clear on auscultation, with a respiratory rate of 46 breaths per minute and no signs of respiratory distress. His heart sounds were normal with a heart rate of 138 beats per minute, pre- and post-ductal oxygen saturations of 100%, a mean arterial pressure of 53mmHg, and bilaterally palpable femoral pulses.
Neurologically, the anterior fontanelle was soft and he was alert but irritable, with a good suck and well-flexed upper and lower limbs. On the remainder of the examination, the abdomen was distended and there was a palpable and tender bladder that was tympanic on percussion. There was no hepatomegaly or spleno-megaly. He had normal male genitalia with a central urethra, no phimosis or paraphimosis, a normal penile length, normal scrotal rugae, bilaterally descended testes and a patent anus with an intact spine. His skin was mildly icteric, but there were no notable rashes. His temperature was 36.9°C.
He was brought to the level 2 neonatal unit for further investigations and management.
Investigations
The baby had a baseline set of investigations performed upon admission, including venous gas, a full blood count, urea and electrolytes, a bone profile, liver function tests, C-reactive protein (CRP) levels, a blood culture, and urine micro-scopy and culture. The results are presented in TABLE 1.
A bedside bladder scan was performed, which showed a largely distended, fluid-filled bladder with no visible obstruction. The baby was subsequently catheterised and his fluid input and output was strictly monitored. The baby was reweighed at 3,120g (5.7% weight loss) and his urine output was 2mL/kg/hour.
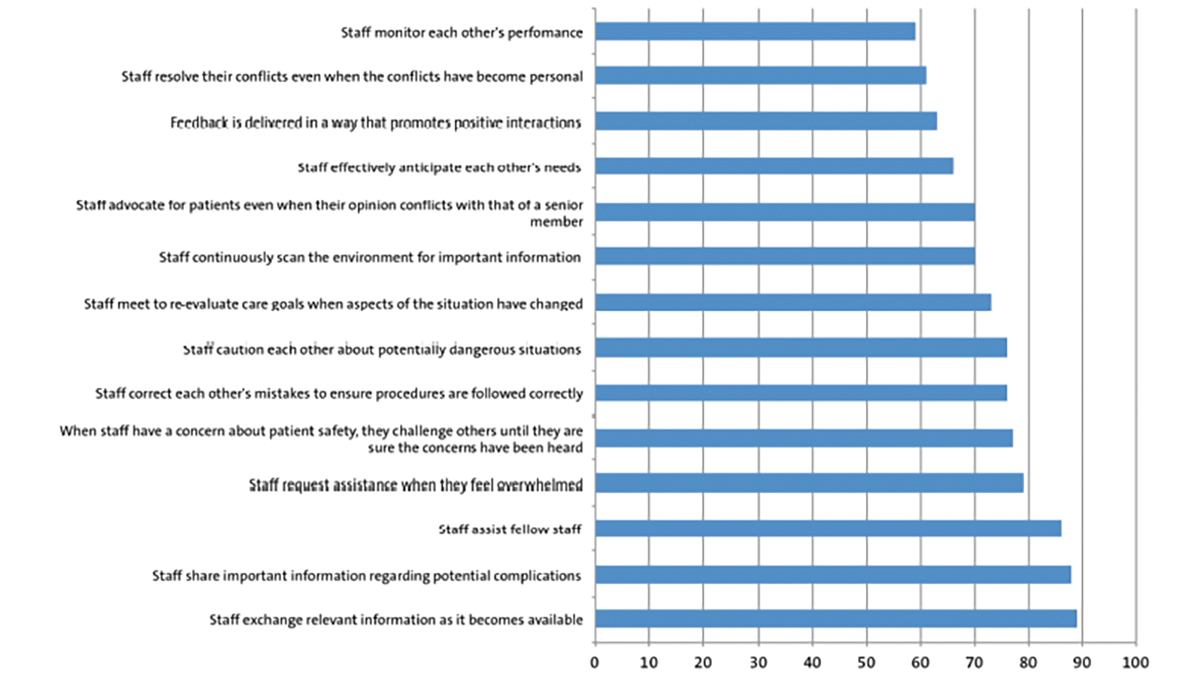
His postnatal USS KUB was expedited and the unilateral right-sided pelvicalyceal dilatation persisted from the antenatal scan but had reduced to 8mm from 15mm. There was also a unilateral right-sided hydroureter. The bladder wall was thick with irregularity and with a hypoechoic bulge at the inferior right aspect of the bladder, initially suspected to be a ureterocele (FIGURE 1).
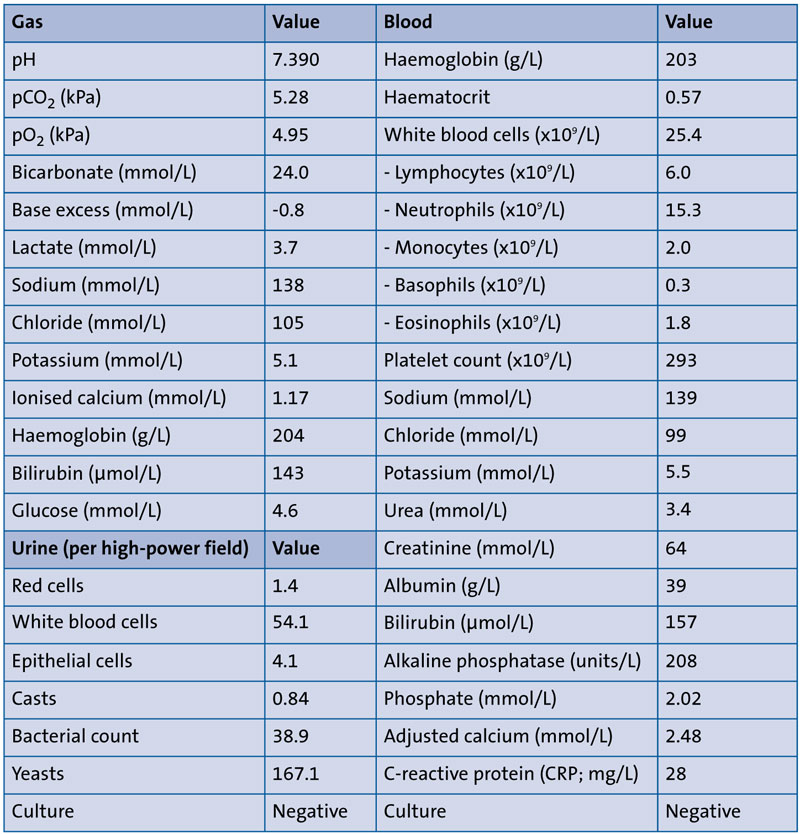
TABLE 1 Results of baseline gas, blood and urine investigations. From a renal perspective, the pH, bicarbonate, urea and electrolytes, and bone profile were all within normal range and the urine culture was negative. The remainder of the initial investigations were unremarkable except for a slightly raised serum CRP and urinary white blood cell count.
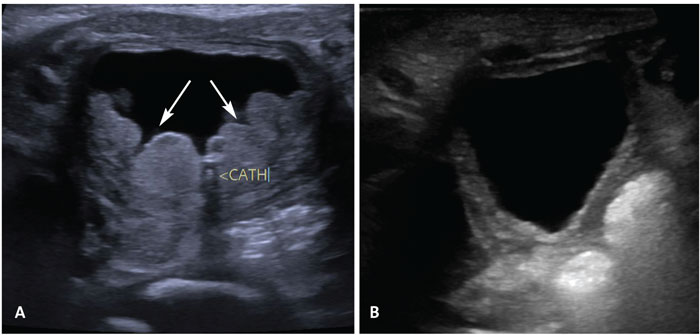
FIGURE 1 (A) The first postnatal ultrasound scan of the kidneys, ureter and bladder (USS KUB) shows a thick bladder wall with hypoechoic bulge projecting into the bladder (directed by the arrows). (B) The final USS KUB during the baby’s admission shows that the bladder wall still remains thick, however the nodular bladder wall mass has almost completely resolved.
Differential diagnosis
In a case of urinary outflow obstruction in a male, the most likely diagnosis is posterior urethral valves (PUV) although in this case the unilateral hydronephrosis contradicted this – children with PUV often have bilateral hydronephrosis. The other potential causes of urinary outflow obstruction are pelviureteric junction obstruction, vesicoureteric reflux, or a ureterocele.
With a CRP of 28mg/L, a urinary tract infection is possible, although this is very unlikely within 24 hours of birth especially in a male, unless there are structural or functional abnormalities. Even so, such abnormalities rarely lead to a urinary tract infection so rapidly.
In terms of rarer differential diagnoses, urinary retention may be caused by mechanical obstruction secondary to tumours or retention secondary to neurological compromise. However, these causes are considered once the more common causes have been ruled out. The baby in this case was unlikely to have prune belly syndrome as, although he had hydronephrosis and bladder enlargement, both testes were descended and the abdomen was not largely and visibly distended, as seen in those with prune belly syndrome.
Treatment
The acute urinary retention was relieved by catheterising the baby with a 6Fr naso-gastric tube, albeit with considerable resistance. Subsequently, 56mL of urine was immediately drained and sent for microscopy and culture.
A cannula was inserted and blood samples were taken for a partial septic screen and baseline investigations, and the antibiotic regimen was escalated to benzylpenicillin 50mg/kg twice a day and amikacin 12mg/kg every 36 hours. This was changed back to prophylactic trimethoprim once the blood and urine culture results came back as negative and CRP levels were down-trending.
The baby was kept nil-by-mouth and commenced on intravenous 10% dextrose with additives at 90mL/kg/day.
We contacted the local paediatric surgical unit and the baby was transferred for further investigations and a surgical review.
Outcome and follow-up
Upon transfer at the paediatric surgical centre, the baby had a micturating cystourethrogram (MCUG), which showed a trabeculated bladder with vesicoureteric reflux extending to the right pelvicalyceal system, with a dilated and tortuous right ureter (FIGURE 2). However, there was no ureterocele, PUV, or pelviureteric junction obstruction seen (FIGURE 2). A repeat USS KUB was performed, which confirmed a posterior trabeculation of the bladder, stable right-sided pelvicalyceal dilatation and hydroureter, however the cause of the urinary outflow obstruction was still unknown.
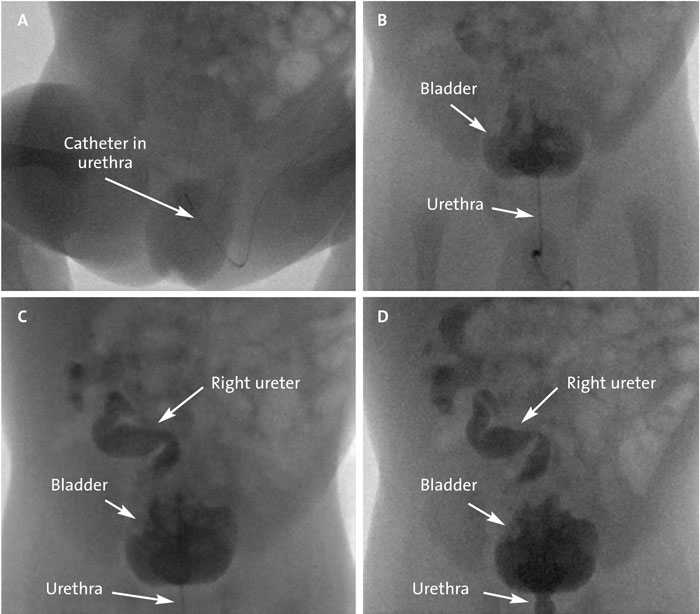
FIGURE 2 The micturating cystourethrogram (MCUG). In images (A) and (B), upon administration of the dye, the urethra can be seen opacifying (with and without the catheter in situ), but there is no overt dilatation of the posterior urethra, particularly post-catheter removal (which contradicts a diagnosis of PUV). In (B), there is evidence of a thick, trabeculated bladder, but no ureterocele. In (C) and (D) it is evident that the right ureter is dilated and tortuous, with right-sided reflux extending into a dilated renal pelvis and blunted calyces – there is no evidence of pelviureteric junction obstruction.
As there was no indication for surgical intervention and the urine output remained good, intravenous fluids were stopped and feeds were reintroduced via a bottle (to monitor fluid input). It was then decided that the catheter should be removed and the baby’s urine output monitored without a catheter with the view to repeating the USS KUB in one week, alongside an ultrasound scan of the spine, to rule out any neurogenic causes of the urinary obstruction. The baby passed urine independently once the catheter was removed and maintained a good urine output and was steadily regaining weight.
The ultrasound scan of the spine was normal, however the repeat USS KUB showed that the nodular mass in the posterior aspect of the bladder seen in the initial USS KUB, had almost completely resolved. This was suggestive of pseudo-tumoral cystitis, with some nodularity near the base, more on the right (FIGURE 1). The pelvicalyceal dilatation had reduced to 5mm but the bladder remained concentrically thick-walled (FIGURE 1).
In light of the diagnosis of pseudo-tumoral cystitis, a urine sample was sent for histopathology, which just found occasional bland urothelial cells, squamous cells, and intact red blood cells (with no casts or malignant cells seen).
Following these investigations and upon reaching the diagnosis, the baby was clinically well; he was feeding well, passing urine without a catheter and passing meconium, and his weight was 3,360g. His urea and electrolytes, bone profile and gases were monitored regularly during his admission and they remained stable. He was discharged home on prophylactic trimethoprim. At three months of age he had a repeat USS KUB, which showed a persistent right-sided hydronephrosis, with a right renal pelvic dilatation of 7mm and right urothelial thickening. Prophylactic trimethoprim was continued. At six months of age, he had another USS KUB, which showed complete resolution of the unilateral hydronephrosis. In the six months since discharge, he has been passing urine independently and has not had any urinary tract infections. He will continue to be followed up and monitored.
Discussion
Pseudotumoral cystitis is a rare inflammatory condition that comes under the umbrella term of eosinophilic cystitis disorders.1 Eosinophilic cystitis is associated with a pathological eosinophilia and can affect any part of the bladder wall and affect its function2-4 In children, boys are more often affected than girls, however across all ages it is equally distributed between both genders.5
The first published cases of pseudo-tumoral cystitis in children were described in 1983, of which one of the three cases described, was in a neonate and is still the only reported case of neonatal pseudo-tumoral cystitis, until now.6 Including these three cases and our case, there have been a total of nine cases of pseudotumoral cystitis in children published in the literature to date.1,6,7 However, there have been a total of 22 reported cases of eosinophilic cystitis in children and in these cases, there were no comments about whether these children were affected by the pseudotumoral form of the disorder.2-4,8-11
Our case of pseudotumoral cystitis presented as urinary outflow obstruction and abdominal pain. This has been a common presentation in the few cases of pseudotumoral cystitis and eosinophilic cystitis reported, as well as urinary tract infection, dysuria, haematuria, and increased urinary frequency and urgency.1,3,6-9,11,12
Due to the paucity of reported cases, some causes of eosinophilic cystitis have been suggested including infection, allergy or a family history of atopy, but there is no robust evidence for this.1,11 In one of the two paediatric cases of eosinophilic cystitis reported by He et al, the child had a history of atopy and both cases demonstrated a marked eosinophilia in the blood and in bone marrow histology.8 In the three paediatric cases of eosinophilic cystitis presented by Redman et al, all had a history of atopy.4 The link between eosinophilic cystitis and atopy has been hypothesised as being due to an antigenic stimulus that provokes an IgE-mediated eosinophilia throughout the bladder wall, resulting in mast cell degranulation and the release of inflammatory mediators.12 In our case, no direct cause was found and there was no family history of atopy. The baby had slightly raised eosinophils; however, the mother’s eosinophils were normal. In the neonatal case of pseudotumoral cystitis reported by Takvorian et al, no cause was found.6
In terms of investigations, we performed a USS KUB, which showed features suggestive of pseudotumoral cystitis. All of the available literature suggests that ultrasound is the best imaging modality for all types of eosinophilic cystitis, however the diagnostic features are not well known.1 Various cases have reported ‘mass-like’ appearances on ultrasound or diffuse mucosal lesions with diffuse and uneven thickening of the bladder wall, which are also often seen on computerised tomography (CT) and MCUG.3,11,12 Shelmerdine et al, found that a colour Doppler ultrasound showed a hyperaemic appearance, suggestive of inflammation.12 The diagnosis can be confirmed by cystoscopy showing an inflamed bladder wall, with a biopsy demonstrating congestion, oedema, and infiltration of blood vessels and eosinophils in the muscular layer alongside focal muscular necrosis.3,12
Unfortunately, there is no definitive treatment to eliminate eosinophilic cystitis and the course of the disease is unpredictable, although usually it is self-limiting.12 The use of antibiotics, steroids and antihistamines has been trialled in children with eosinophilic cystitis with variable success.3,7-9 Often in cases where the inflammatory mass is resistant to medical management, resection has been performed and a cystectomy can be considered.3,11 In our case, antibiotics were administered, which perhaps helped to reduce the size of the obstructive inflammatory bladder mass; hence, this may explain why the baby was independently passing urine without a catheter after a few days of antibiotic therapy.
Conclusion
This article presents an interesting and rare cause of urinary outflow obstruction in a male infant, which is something for neonatologists and paediatric surgeons to consider as a differential diagnosis when common causes of obstruction have been ruled out. The infant presented at 24 hours of age with urinary retention. He was catheterised and started on antibiotics due to raised C-reactive protein. The initial ultrasound scan showed a hyperechoic bulge at the base of the bladder with persistent hydronephrosis, resolving on repeat ultrasound. Due to the paucity of cases in the paediatric population, particularly in neonates, the long-term clinical course or complications arising from pseudotumoral cystitis are not known. Therefore, it would be useful for such cases to be followed closely over a period of time with findings published.
To this day, we can confirm that the baby in our case is still wetting nappies and there have been no reports of urinary tract infections or clinical signs of atopy. Therefore, we hope that cases of pseudotumoral cystitis will be a single event in a child’s life with no impact on their future health.
Author contributions, acknowledgement and
parental consent
Dr Arameh Aghababaie conducted the data analysis, research and writing of the article. Dr Hesham El Sayed had the idea for the article and edited the article. The authors would like to thank Mrs Ashwini Joshi for her contribution to the urological and surgical care of this infant. Verbal consent for publication was obtained from the parents of the patient.
Or read this article in our
Tablet/iPad edition
- All parents should be advised to seek urgent medical attention if their baby has not passed urine within 24 hours of birth, especially those who are more at risk due to antenatal renal abnormalities.
- Acute urinary retention in a newborn is a surgical emergency and needs to be managed urgently.
- Posterior urethral valves are the most common cause of urinary outflow obstruction in male infants; however, when it is not clear it is important to consider the possibility of an obstructing mass or retention secondary to neurological causes.