Volume guarantee ventilation in a NICU: a review of practice
ST7 Paediatrics
femi.adeniyi@nhs.net
Chukwuemeka Kenechukwu
Senior Clinical Fellow
Sanjeev Rath
Consultant Neonatologist
Hossam Khaliel
ST2 Paediatrics
Neonatal Unit, Wirral University Teaching Hospital
Bronchopulmonary dysplasia (BPD) is a chronic lung disease (CLD) of primarily premature infants resulting from an imbalance between lung injury and repair in the developing immature lung. The pathogenesis is complex and determined by the interactions between the ventilator settings and patient-related factors.1
Volume guarantee ventilation (VGV) results in more consistent tidal volumes that allow automatic weaning of airway pressure. Volume guarantee use has increased in neonatal practice due to potentially significant findings compared to the regular pressure-limited ventilation mode, reducing inadvertent hypocarbia (hypocapnia)2,3 and periventricular leukomalacia (PVL).4,5
VGV, also known as pressure-regulated volume control ventilation, is characterised by the delivery of pressure-controlled inflation using a decelerating flow pattern. The end of inspiration is time-cycled. The operator sets a target tidal volume for these pressure-controlled inflations. After each mechanical inflation, the delivered tidal volume is measured, and the machine (in our setting, GE Healthcare Engstrom Carestation and Fabian ventilators) adjusts the peak pressure to achieve the set tidal volume.6,7 Hence the term ‘guarantee’ is not accurate as the delivered tidal volume is not guaranteed but stabilised as much as possible.
The excessive tidal volume is more closely associated with ventilator-induced lung injury (volume trauma) than excessive end-inspiratory pressure.8,9 Hence the increasing drive towards volume targeted ventilation rather than pressure.
In this article, we share our experience of outcomes in our most vulnerable preterm babies (≤26 weeks’ gestation) over a 12-month period (May 2019 to April 2020) since introducing VGV into our neonatal intensive care unit (NICU) at Wirral University Teaching Hospital.
Objective
To review the incidence of CLD at 36 weeks post-menstrual age and severe intraventricular haemorrhage (IVH) and/or PVL in extreme preterm infants of ≤26 weeks’ gestation.
Methods
A retrospective comparison of the incidence of CLD and grade 3-4 IVH/PVL in a cohort of babies born at 22-26 weeks’ gestation on the mechanical ventilation volume guarantee (VG) mode versus SIMV-PC mode (synchronised intermittent mandatory ventilation – pressure controlled). The secondary outcome measures were differences in the use of dexamethasone and diuretics in the two cohorts.
A total of 74 eligible babies were identified from BadgerNet over a two-year period (ie babies born at ≤26 weeks’ gestation and requiring ventilation). We compared data from the VG group (May 2019 to April 2020; 31 babies) with the SIMV-PC group (May 2018 to April 2019; 43 babies).
Results
Twelve babies died in the study period: eight babies in the SIMV-PC cohort and four in the VG.
CLD incidence
Babies on the SIMV-PC mode of ventilation had a CLD incidence (ie the baby required non-invasive ventilation at 36 weeks post-menstrual age) of 88.6% (31 out of 35 neonates) compared to 70.4% (19 out of 27) of babies receiving the VG mode of ventilation (FIGURE 1).
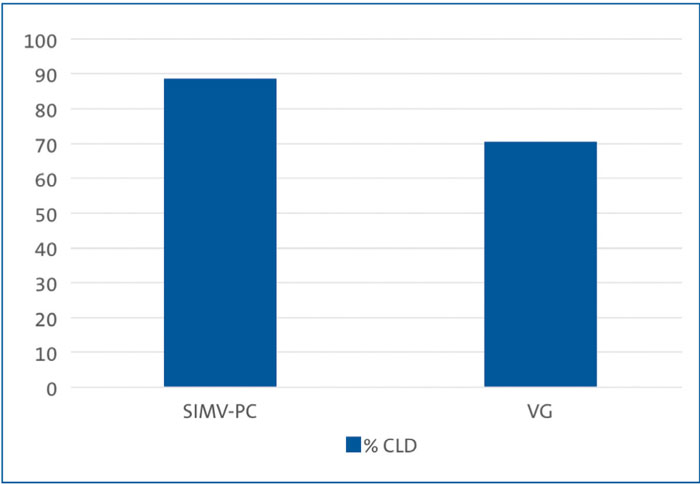
FIGURE 1 Comparing the incidence of CLD in preterm babies on the two modes of ventilation.
IVH/PVL
21% of babies that received SIMV-PC ventilation had severe IVH/PVL (grades 3-4) while 22.5% of babies on the VG mode of ventilation had severe IVH/PVL.
67% of the babies that received SIMV-PC and had severe IVH/PVL had it on the day 1 scan. 43% of babies on the VG mode of ventilation who had severe IVH/PVL had IVH on the day 1 scan. Consequently, we were unable to quantify the occurrence of ventilation-associated IVH/ PVL in the two groups as many of the babies in the two cohorts had bled on day 1.
Secondary outcome measures Dexamethasone to treat evolving BPD was used in 74% (26 out of 35) of neonates in the SIMV-PC cohort and 52% (14 out of 27) in the VG cohort. Diuretic use was 63% (22 out of 35) with SIMV-PC and 85% with VG (23 out of 27; FIGURE 2).
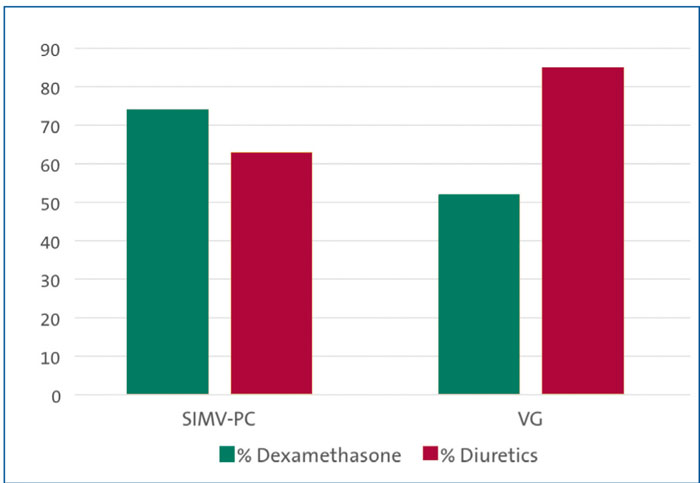
FIGURE 2 Secondary outcome measures: use of dexamethasone and diuretics.
Conclusion
We are pleased to see that our most vulnerable preterm infants (≤26 weeks’ gestation) that received VGV had a lower incidence of CLD when compared to the SIMV-PC cohort. Our findings align with evidence from various meta-analyses, supporting the VG mode of ventilation in neonates.
We have seen a decrease in the use of steroids since starting using VGV. The use of diuretics in the VG group increased but we can not ascertain if this is associated with the reduction in incidence of CLD.
Based on our short study, we would encourage neonatal units to consider adopting the VG ventilation strategy.
Or read this article in our
Tablet/iPad edition