The role of procalcitonin when reviewing antibiotics for possible early onset infection
Deciding when to stop antibiotics in babies being treated for possible early onset infection can be challenging. This article is a systematic review of the role of procalcitonin as an aid to clinical decision making in these cases. The review was designed to investigate whether low procalcitonin levels at 24-48 hours of life, in apparently well term neonates, can enable clinicians to safely stop antibiotics in the absence of a positive blood culture.
Charlotte R. BurleighSpecialty Trainee 5, Bradford Teaching Hospitals NHS Foundation Trust, charlotte.burleigh@nhs.net
Katherine J. Pettinger
Specialty Trainee 6, Bradford Teaching Hospitals NHS Foundation Trust
Early-onset neonatal sepsis is a rare but serious condition, with a non-specific presentation. Recognising this, many apparently well newborn infants are proactively commenced on antibiotics shortly after birth based on risk factors or clinical indicators associated with neonatal infection.1
Once the decision has been made to start antibiotics, the next challenge is deciding when it is safe to stop. While it is important not to undertreat truly septic infants, clinicians also have a responsibility towards antibiotic stewardship. Additionally, with prolonged antibiotic courses comes increased length of stay, more opportunities for medication errors and the discomfort and infection risk associated with repeated intravenous cannulation.
In the UK, antibiotic courses are typically reviewed at 36 hours taking into consideration the clinical history, examination, blood culture and C-reactive protein (CRP) levels.1 The yield of positive blood cultures from newborns is recognised to be low, meaning that a negative blood culture alone is often not reassuring enough to discontinue treatment. In clinically well infants with negative blood cultures, CRP at 18-24 hours can become the key decision-making factor. While low results (typically <10mg/L) are generally felt to be reassuring and very high results (eg >30mg/L) warrant further review, there is a ‘grey area’ that could lead to inconsistencies in practice. Procalcitonin (PCT) might have a role in such cases.
PCT, a precursor of the hormone calcitonin, is a 116 amino acid peptide.12 PCT production is thought to be induced in response to bacterial lipopolysaccharides and pro-inflammatory cytokines, eg interleukin-6.3 PCT has a short half-life and levels have been shown to rise rapidly in response to systemic bacterial infections and correlate with illness severity and mortality in septic patients.2-4 PCT levels in the neonatal population are more challenging to interpret due to a physiological rise in PCT in the first 48 hours of postnatal life (peak around 24 hours).4-6 This means that, if PCT is to be utilised as a marker of early onset neonatal infection, cut-off values will be age and time dependent.6 The optimal timing of PCT testing in newborns and the reliability of PCT levels before the physiological 24-hour peak has not been clearly established. This review therefore seeks to establish whether PCT is a useful test once antibiotics have been started for suspected infection.
Methods
Searches were carried out by the authors independently. The inclusion criteria were:
- full text articles
- written in English language
- review articles including original data, or original articles
- commentaries, editorials, case reports and case series were excluded.
The exclusion criteria were:
- non-English language
- duplicated articles
- not relevant to the clinical question.
Cochrane library
Search: pro-calcitonin or procalcitonin in title, abstract or keyword. Search limited to Cochrane groups ‘neonatal’ and/or ‘child health’. This search produced 15 results, five of which were relevant and selected for full text review.
Medline
The search terms used in Medline (Ovid MEDLINE(R) ALL 1946 to November 25, 2020) were:
- infant/ or infant, newborn/ or neonat*.mp
- procalcitonin.tw or exp Procalcitonin/ or pro-calcitonin.tw or PCT.tw
- neonatal sepsis/ or neonatal sepsis.tw or neonatal infection.tw or early onset sepsis.mp or early onset infection.mp
- #1 AND #2 AND #3.
This search produced 202 results of which 44 papers were selected for full-text screening based on the authors’ inclusion criteria.
Results
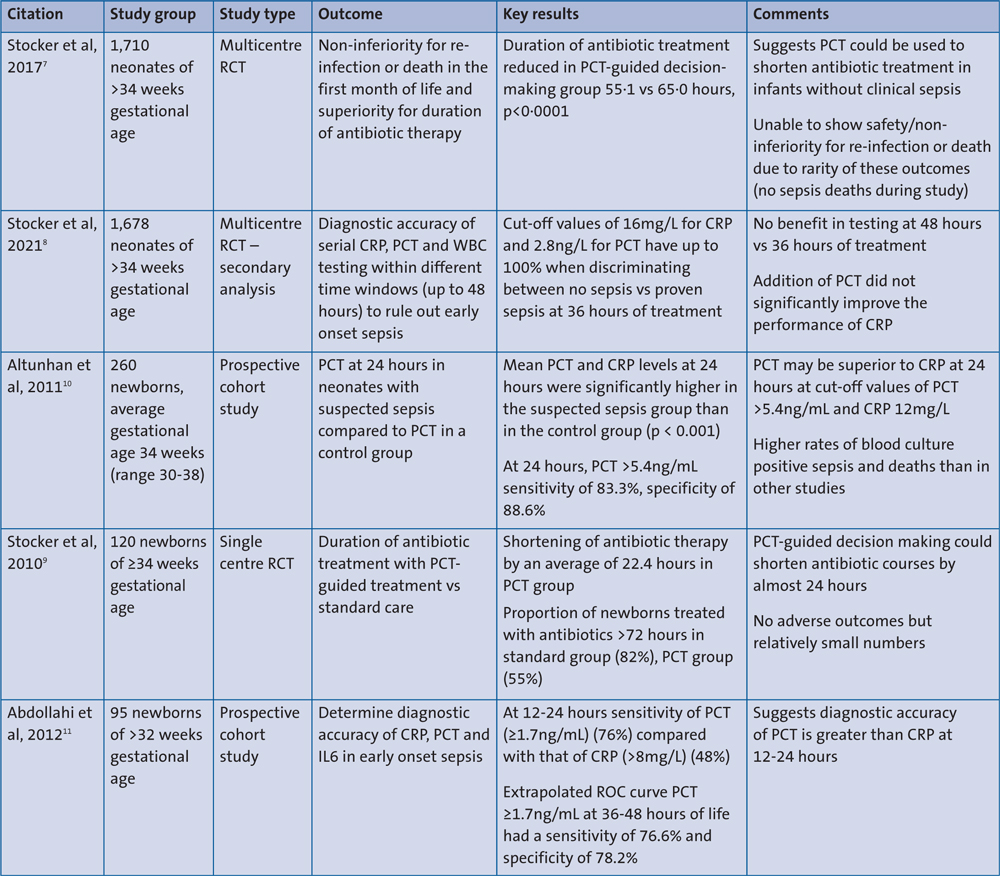
Full-text screening yielded five studies that were relevant to the question. All relevant papers obtained through the Cochrane database were also found by the Medline search. The results are summarised in TABLE 1.
TABLE 1 Summary of evidence. Key: RCT=randomised controlled trial; CRP=C-reactive protein; PCT=procalcitonin; WBC=white blood cells; IL6=interleukin-6.
Discussion
The largest study evaluating PCT as a decision-making tool to guide antibiotic courses for early onset infection is the Neonatal Procalcitonin Intervention Study (NeoPInS).7 NeoPInS was a multicentre randomised controlled trial involving term and late preterm (>34 weeks’ gestation) neonates with suspected early onset infection. PCT-based decision making involved antibiotics being discontinued when two consecutive PCT values were within normal range (up to 10ng/mL at 18-36 hours of life). Crucially, PCT-based decision making was only applied to infants where sepsis was felt to be ‘possible’ or ‘unlikely’. These categories included infants with negative blood cultures and either one or two ‘abnormal findings’, eg infection risk factors, abnormal laboratory tests or clinical symptoms. The study threshold for an abnormal CRP was >10mg/L. NeoPInS suggested that in the lower-risk group of infants, such as the one in our clinical scenario, using PCT resulted in shorter antibiotic courses (55·1 vs 65·0 hours, p<0·0001). The authors conclude that PCT-based treatment was superior to standard care.
A secondary analysis of NeoPInS suggests that thresholds of CRP at 16mg/L or PCT at 2.8ng/mL can discriminate between proven sepsis (positive blood culture) and no sepsis with up to 100% sensitivity at 36 hours.8 When infants with ‘probable sepsis’ (sepsis features but negative blood cultures) were grouped with proven cases, CRP at 13mg/L or PCT at 1.5ng/mL were found to give a sensitivity of approximately 95%.
This paper suggests that combining PCT with CRP does not increase the overall sensitivity and specificity. This does not necessarily mean that combining PCT and CRP is of no value. Positive predictive values for PCT and CRP were not obtained in this analysis due to the small number of proven sepsis cases. Importantly, this analysis demonstrated that the negative predictive value of CRP and PCT did not change significantly between 36-48 hours. This suggests that there is no benefit to waiting until 48 hours to make treatment decisions based on laboratory results. Given that commonly used antibiotics such as benzylpenicillin are usually prescribed 12-hourly, decision making at 36 hours potentially saves at least one dose of antibiotics and could potentially save a further night in hospital.
A similar, smaller prospective interventional study by the same group of authors found an average reduction of 22.4 hours in the antibiotic treatment duration in their PCT-guided group compared with standard care.9 Of note, they concluded that PCT was most likely to influence management in infants with a CRP >5mg/L at 24 hours of antibiotics treatment. This is reflected in a more significant reduction in the proportion of infants treated for >72 hours in the PCT group (57%) compared with the standard group (95%) (odds ratio 0.076; 95% confidence interval: 0.016-0.359). Both this study and NeoPInS were not able to demonstrate non-inferiority in terms of mortality and reinfection. Despite relatively large sample sizes, the rarity of both death and reinfection meant that the authors were unable to make confident assertions about the safety of PCT-guided treatment. No sepsis-related deaths occurred across either study group.
Altunhan et al suggest that PCT may be a more reliable marker of infection than CRP at 24 hours.10 This observational study found PCT at 24 hours was significantly higher in a group of neonates with probable sepsis compared to a group with mild features or isolated sepsis risk factors. However, the population in this study had a very high rate of positive blood culture (40%) and included babies <34 weeks’ gestation, therefore it was not considered in further detail, as it was not representative of the group of babies we were focussing on.
Abdollahi et al evaluated PCT as a diagnostic tool, comparing it with CRP and interleukin-6.11 This study included both apparently well infants with maternal risk factors for infection and symptomatic babies, with a gestation range of 32-38 weeks. Blood for PCT was taken at 12-24 hours after admission (all babies were admitted within 12 hours of birth). CRP and PCT were found to be correlated in infants with blood culture-positive sepsis (correlation coefficient, r=0.54, p<0.001). The sensitivity of CRP (cut-off >8mg/L) at 12-24 hours was increased from 48 to 82% with the addition of PCT (cut-off ≥1.7ng/mL). While this suggests that PCT could be a useful adjunct to CRP, based on National Institute for Health and Care Excellence (NICE) guidance, a single CRP would rarely be taken and interpreted at 12 hours of life. It is, therefore, difficult to know how useful this result is in practice. The authors’ extrapolated receiver operating characteristics (ROC) curve suggested that PCT ≥1.7ng/mL alone at 36-48 hours of life had a sensitivity of 76.6% and specificity of 78.2%.
Conclusion
These studies suggest that PCT testing has the potential to guide decision making in suspected early onset neonatal infection. The heterogeneity of studies in this area, including gestation, definitions of sepsis, timing of samples, and cut-off values for associated inflammatory markers, mean that reviews and meta-analyses are limited. It is therefore difficult to translate these results into clinical context. Further large-scale studies may help to reassure clinicians about the safety of PCT-guided decision making, as well as determine appropriate cut-off values. Additional laboratory tests come with financial implications and cost-effectiveness should be considered. The laboratory cost of PCT testing is currently around £15 and some protocols recommend at least two PCT tests (including NeoPInS). While this is not insignificant, the potential savings in terms of reducing antibiotic administration and costly prolonged hospital stays for even a small proportion of infants may outweigh this. The inclusion of a formal cost-effectiveness analysis would be beneficial in future studies.
Contributions
The concept for the article was devised by CB. Both authors planned the methods together. The literature search was carried out by both authors independently. The paper was written by both authors collaboratively.
Or read this article in our
Tablet/iPad edition
- Decision making about antibiotic treatment should not rely on any single blood test.
- Procalcitonin (PCT) is a potentially useful adjunct to C-reactive protein (CRP) levels in term babies being treated for suspected early onset neonatal infection.
- Low PCT and CRP results should allow clinicians to be more confident in their decision to stop antibiotics in apparently well babies.
- PCT results need to be interpreted according to the time they are taken due to physiological variations in PCT after birth.