Problems and perceptions around severe bronchopulmonary dysplasia
Advances in neonatal care have led to increased survival of the extreme preterm infant, yet bronchopulmonary dysplasia (BPD) as a comorbidity has shown an increasing trend. A major proportion of preterm infants who remain invasively ventilated at term-corrected gestational age go on to require long-term respiratory support yet the management and prognosis for severe BPD shows wide regional variation depending on availability of resources. This article addresses conundrums regarding the management of severe BPD and the care pathway, including a survey of practice to give a UK regional perspective.
Amitava Sur1amitava.sur@elht.nhs.uk Omendra Narayan2 Anshuman Paria1 Stuart Wilkinson2 Savi Sivashankar1 Andrew Cox1 Jasim Shihab1 1Consultant Neonatologist, Lancashire Women and Newborn Centre
2Consultant Respiratory Paediatrician, Royal Manchester Children’s Hospital
The magnitude of the problem
The outcome of infants born extremely preterm has steadily improved over the years. Since 2006, the reported survival at one year of infants born between 23+0 and 23+6 weeks’ gestation in the UK was 38%. In a National Neonatal Research Database (NNRD) study, the annual percentage change of survival of the extremely preterm infant was found to be greatest at 22+0 to 23+6 weeks (6.03%).1 With a greater understanding of management strategies, the incidence of comorbidities associated with extreme prematurity has also reduced, with the exception of BPD,2 which has often showed a steadily increasing trend. In a recent meta-analysis the global incidence of BPD was reported to be anywhere between 10-89% with 47% in the UK, which was one of the highest reported in Europe.2 Despite therapeutic refinements, this rate of steady increase might reflect a more complex multifactorial aetiology and raise concerns for the considerable impact on infrastructure.
The neonatal care model in the UK is delivered through operational delivery networks (ODN) based on geographical areas of the clinical senates. Each ODN comprises neonatal intensive care units (NICU), local neonatal units (LNU) and special care (baby) units (SCBU or SCU). Among the NICUs, depending on geographical location, one or more will be a tertiary referral centre with provision for specialised services such as respiratory medicine, surgical principles, etc, and the rest will be tertiary units based on district general hospitals.
With improved expertise in caring for the extremely preterm infant, the survival and mean hospital stay of such infants has increased. It is almost always the case that an infant born at the extremes of gestational age would be on some kind of respiratory support at term-corrected age and often invasively ventilated. The average number of days spent in the NICU for such infants has been reported to be over 100, with around a third discharged home on oxygen.3 It is important to note that many are not discharged from the NICU to home directly. These babies are transferred for ongoing respiratory care to a paediatric intensive care unit (PICU) or high-dependency setting for joint care under the paediatric respiratory team. In fact, a major subset of children on long-term ventilation (LTV) in paediatric units is constituted by ex-preterm infants with severe BPD. A recent review undertaken for the Paediatric Critical Care Clinical Reference Group4 showed that the average length of hospital stay from the time of identification of the need for LTV to discharge home is an average of 7-9 months implying long-term bed occupancy. The ripple effect thus created leads to admission refusals to paediatric intensive care and often transfers out of the network for patients, exerting significant strain on health economics.
Defining BPD
Definitions of BPD are inconsistent but the following categories have been proposed:5
- mild BPD: infants who received oxygen or respiratory support for >28 days but were on room air at 36 weeks’ corrected age for a preterm infant
- moderate BPD: infants who required supplemental oxygen at <30% fraction of inspired oxygen concentration (FiO2) at 36 weeks’ corrected age
- severe BPD: the requirement of >30% oxygen or positive pressure at 36 weeks’ corrected age.
BPD is predictive of respiratory outcomes during the first 1-2 years of life. Oxygen and/or respiratory support at 40 weeks postmenstrual age best predicts ongoing respiratory morbidity at 18-24 months of age.5
Beyond statistics: quality of life for caregivers and staff morale
The prospect of ongoing care, acute episodes and uncertainty of outcome often weighs heavily on the parents and medical staff looking after these infants. In an observational study it was found that very preterm birth was itself a risk factor for impaired quality of life (QoL) among parents.6 It was found that 20% of mothers had clinically significant levels of depression whereas 43% had moderate to severe anxiety. Parental role alteration (P<0.01) and prolonged ventilation (P<0.05) were associated with increased depressive symptoms.7 Another study that aimed to assess the impact of BPD directly on health related QoL (HRQoL) of caregivers found an association between lower HRQoL scores and caregivers of BPD children with frequent respiratory symptoms and acute care usage.8 Much less reported but equally pertinent is the anxiety and sense of futility among medical staff, especially the nursing team. The emotional support around severe BPD is an aspect of care we acknowledge but often do not act on. The presence of an in-house clinical psychologist or staff support system is not consistently available across all NICUs in the UK.
Interdisciplinary liaison and challenges to service delivery
Fundamentally, an infant diagnosed with severe BPD will require input from a number of specialist teams including the respiratory team, paediatric intensive care team, paediatric cardiology, gastroenterology, dietitians, clinical pharmacists, physiotherapists and play therapists. In their position statement on interdisciplinary care of children with severe BPD, Abman and colleagues9 outline the evolving journey and complex care needs of these children, stressing the team approach for a better outcome. In reality, this becomes difficult for neonatal units that are not based at tertiary referral centres. The dynamic needs and multiple review points are not adequately met by the local team. However, transition of care to the tertiary referral centres is impeded by regional bed constraints. The coding of care delivery also factors into this process. Of the severe BPD cohort, a majority will be on non-invasive ventilation (NIV) and a minor percentage on invasive ventilation. NIV delivery is coded as high dependency (HDU) while the latter is coded as intensive care (ICU).
The geographical infrastructure of critical care is an important determinant of this transition of care. For example, in the UK, the South West has enhanced the capacity of regional hospitals to deliver paediatric NIV, thus reducing the burden of HDU care on hospitals with a PICU.10 In a large retrospective analysis on paediatric critical care admissions in the UK between 2003 and 2017, it was shown that there was a sharp rise in HDU admissions over the time period, especially in hospitals without a PICU. The data also showed that within the cohort, children requiring HDU and ICU were more likely to have been born preterm and weighed less than 1,500g at birth.11 Regional variations are important in this context for analysis, commissioning and reviewing the care model.
Clinical implications and technical challenges for neonatal units
A detailed discussion about the pathophysiology of BPD is beyond the scope of this article, but it is important to underline that lung physiology evolves with longer stays and persistent inflammation and requires changing strategies when providing respiratory support. The low compliance, low resistance lung of a preterm infant in the immediate postnatal period changes to a high compliance, high resistance lung with heterogeneity and evolving BPD. This often mandates using high volume (use of larger tidal volumes between 8-12mL/kg) and lower rates while ventilating such children with a consistently high positive end-expiratory pressure (PEEP; 8-10cmH2O).12 Neonatologists are often beyond their usual comfort zone using such ventilation strategies. Existing systemic comorbidities necessitate a more holistic viewpoint towards their care. TABLE 1 outlines the clinical challenges and support required in the different domains.
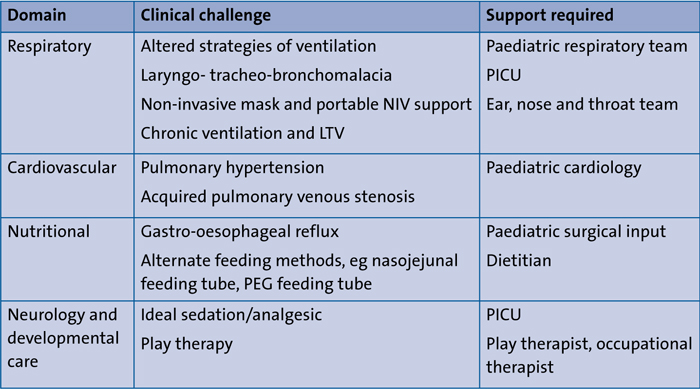
TABLE 1 Systemic comorbidities and multidisciplinary input required for severe BPD. Key: NIV=non-invasive ventilation; LTV=long-term ventilation; PICU=paediatric intensive care unit; PEG=percutaneous endoscopic gastrostomy.
It is sometimes just resource limitation and lack of on-site tertiary services that ails neonatal units. Most infants with severe BPD require non-invasive support with high pressures for extended periods. This becomes challenging as the usual NIV modalities used in the NICU (continuous positive airway pressure therapy, CPAP; bilevel positive airway pressure, Bi-PAP; and high flow nasal cannula therapy, HFNC) do not deliver such high pressures and non-invasive positive pressure ventilation (NIPPV) is still not widely used among neonatal units in the UK. Conventional neonatal ventilators and circuits lack the appropriate interface required for bigger infants. This leads to inadequate ventilation and impaired oxygenation – a combination that leads to worsening of the existing pulmonary hypertension. Use of NIV requires judicious use of sedative/anxiolytics and environmental considerations. The sleep patterns of these children tend to regularise with age and being in a busy NICU can impair their quality of sleep. Supported by respiratory devices that depend on a wall supply of oxygen also means that they cannot be optimally mobilised, leading to poor developmental outcome. Most NICUs in the UK are not kitted out for delivering a chronic care model.
Perceptions of neonatologists regarding management challenges: a survey
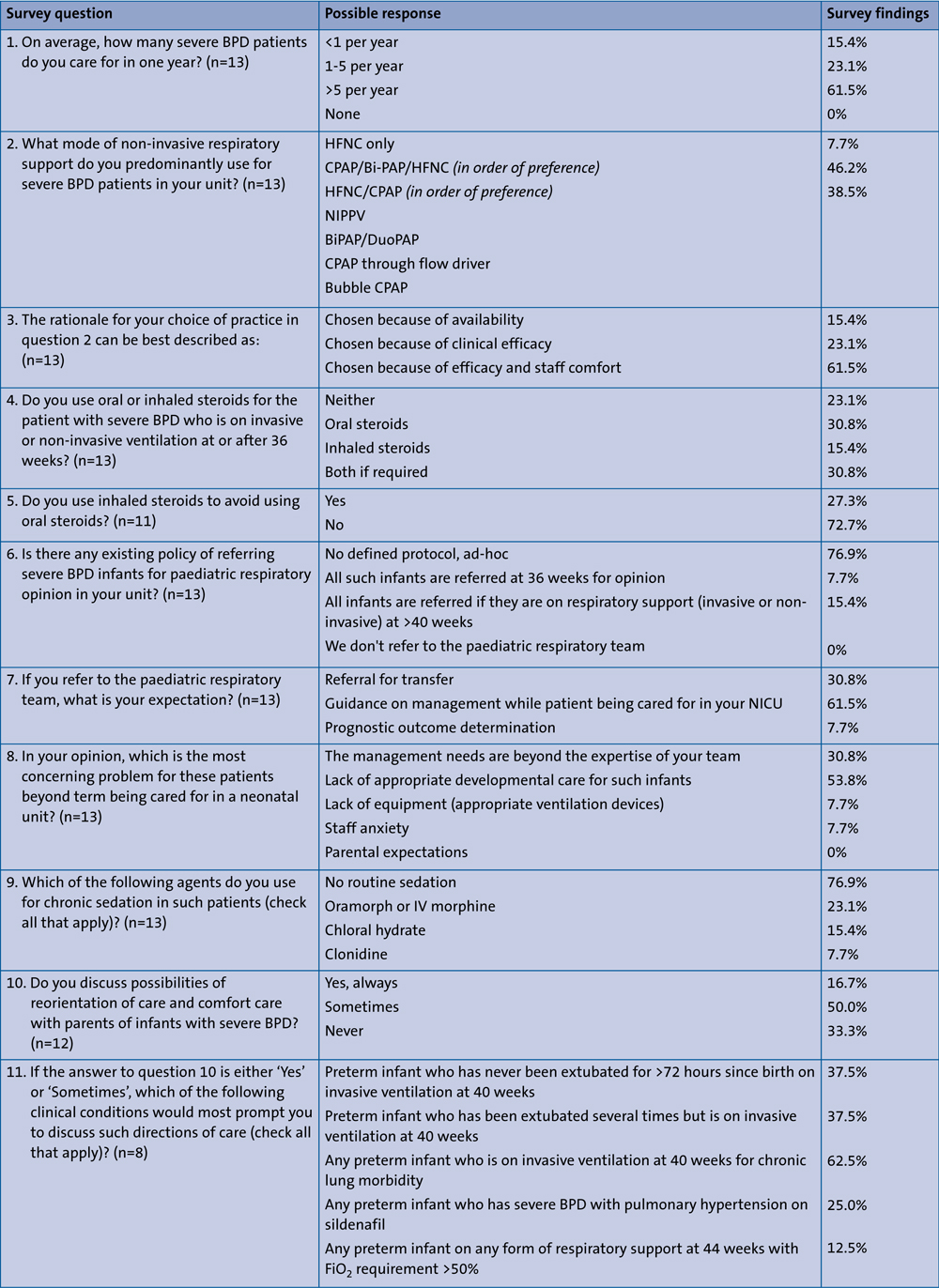
The authors conducted an online questionnaire survey among the neonatal units in the North West (NICUs and LNUs) addressing certain conundrums regarding management of severe BPD and the care pathway. The questions asked, the options provided and the survey findings are detailed in TABLE 2.
TABLE 2 Practices around care of the infant with severe BPD: survey questions and possible responses.
The obvious trends that emerged from that survey were as follows:
- Most neonatal units care for five or more babies with BPD per year.
- The most commonly used modalities of respiratory support are CPAP and HFNC and only a very few units use NIPPV. This choice is guided by efficacy and staff comfort.
- The process of referral to the respiratory team is not guided by any age cut-offs and is ad-hoc (FIGURE 1). The expec-tation is guidance on management and ultimately transfer of care (FIGURE 2).
- Most units believe that after 44 weeks, management is beyond the clinical expertise of the neonatal team and developmental care is inappropriate (FIGURE 3).
- Most neonatologists would discuss direction of care for severe BPD patients and that would be guided by the presence of invasive ventilation and/or pulmonary hypertension requiring therapy.
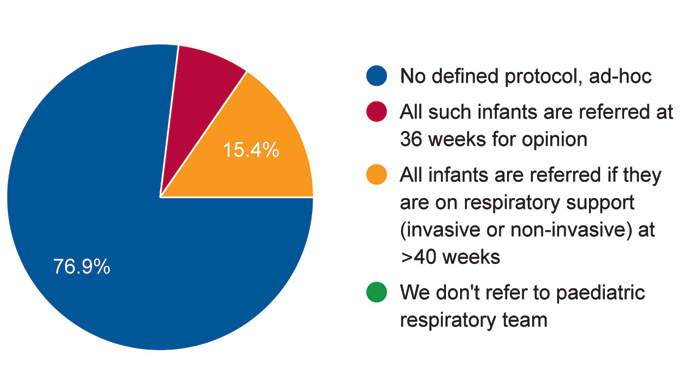
FIGURE 1 Survey responses to the question: Does your unit have an existing policy for referring infants with severe BPD for paediatric respiratory opinion?
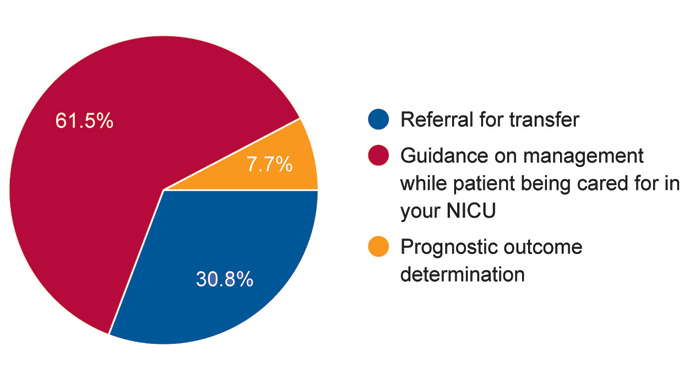
FIGURE 2 Survey responses to the question: If you refer to the paediatric respiratory team, what is your expectation?
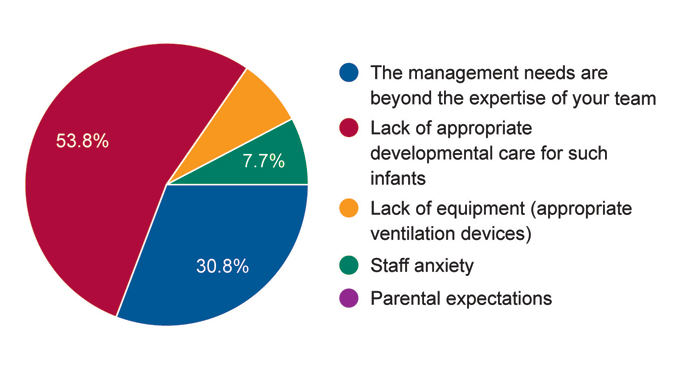
FIGURE 3 Survey responses to the question: In your opinion, which is the most concerning problem for these patients beyond term being cared for in a neonatal unit?
Assessment for LTV: tracheostomy or home CPAP
A major proportion of infants who remain invasively ventilated at and after term gestational age would go on to require long-term respiratory support, either in the form of home CPAP/Bi-PAP through a mask or tracheostomy. This assessment is always made by the paediatric respiratory team and follows a complex individualised algorithm. As a rule of thumb, CPAP at home is preferred only when the infant is able to maintain saturations and remain stable on only low flow oxygen for eight hours or more during the day – the so-called ‘nights and naps support’.13
Although some evidence favours early tracheostomy for better neurodevelopmental outcomes, the logistics are more complicated. In the UK, it involves shifting of the funding process from specialist commissioning to local commissioners, training parents and local carers and arranging for appropriate housing.
This can be a very long and drawn-out process. Hence, as a practical approach, tracheostomy is reserved for those severe BPD patients who are not successfully extubated from invasive ventilation after establishing developmental prognosis and parental understanding.
Searching for achievable solutions
Virtual forum to discuss cases and standardise care and patient flow
Instead of ad-hoc referrals made to specialist teams, each of these severe BPD infants might benefit from a standardised approach to care guided by the tertiary respiratory team. In the authors’ opinion, every infant on invasive or non-invasive support at term corrected age should be a part of a multidisciplinary team (MDT) discussion. This can be facilitated through virtual care meetings.
Developing local expertise
Given the inevitability of the scenario, it would seem that neonatal units require familiarising with the model of chronic care. This would require special interest groups, allied health professionals and additional funding.
Conclusions
Severe BPD is a conundrum arising from the increased survival of extreme preterm infants. Although healthcare professionals are getting better at the initial management of preterm infants, there are voids in the care model for these complex cases and it would seem that a piecemeal approach is often provided rather than a holistic one.
UK tertiary neonatal units based in district general hospitals without HDU facilities face a unique set of challenges posed by limitation in technical knowhow, delay in referral services and increasing staff anxiety, which often lead to suboptimal outcomes.
The importance of developing a chronic care model in neonatal units is a high priority given the increasing pressure on PICU/HDU beds. Dedicated training is required for delivering such care. Early MDT alliance, fostering special interest leads/groups locally and dedicated forums aimed at standardising chronic respiratory care across networks might be the ideal initial steps.
Acknowledgements
The work was conceptualised from a multidisciplinary collaboration between the neonatal team at Burnley General Teaching Hospital and the paediatric respiratory team at Royal Manchester Children’s Hospital to identify gaps in care delivery for patients with severe chronic lung disease. AS and ON drafted the manuscript. AS, ON, AP and SW contributed to proofreading. SW, ON, SS, JS and AC contributed to background information and regional statistics. AS finalised the submitted draft.
Or read this article in our
Tablet/iPad edition
- Severe BPD is a challenging condition arising from increased survival of extreme preterm infants.
- Developing a chronic care model in neonatal units is a high priority for these complex cases.
- Isn’t it time we standardised early multidisciplinary input to help streamline care?