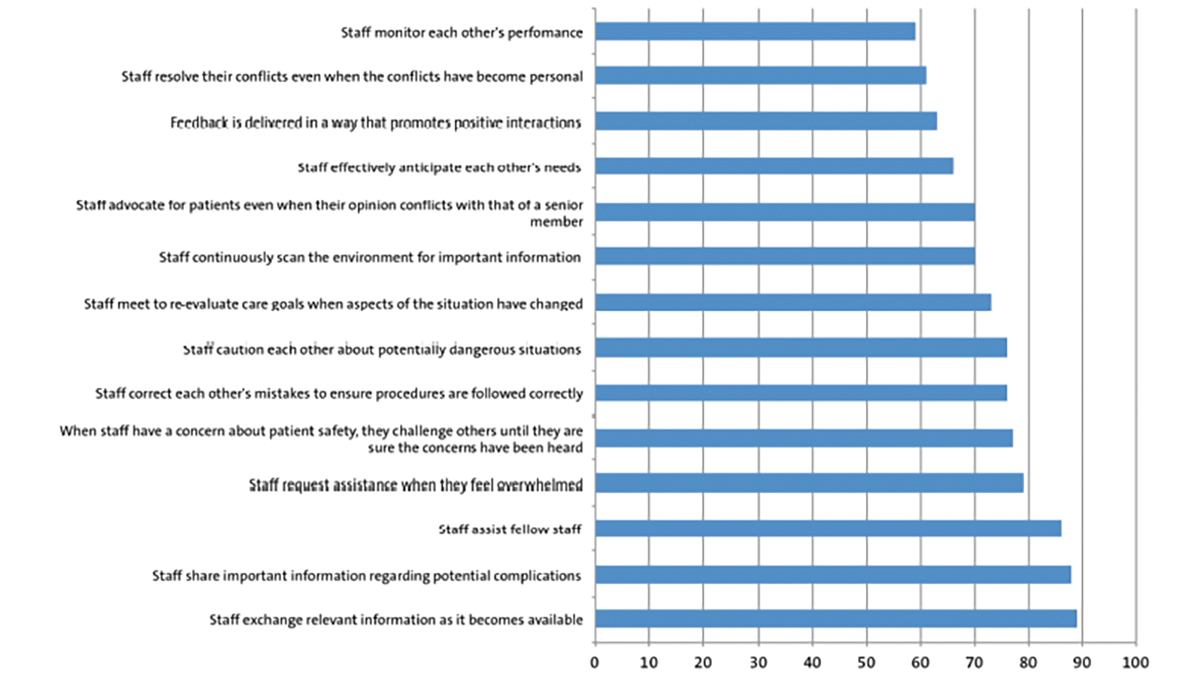
Nocardial pneumonia in an extremely preterm infant
We describe a case of nocardial pneumonia due to Nocardia farcinica in a preterm infant of 25 weeks’ gestation, which is an extremely rare diagnosis in this age group. The infant was subsequently diagnosed with possible chronic granulomatous disease, a known risk factor for this infection. To our knowledge, this case represents the first report of an in-hospital presentation of nocardial pneumonia in an extremely low birthweight neonate.
Wail SeleemMD, Section of Neonatology
Michael Narvey
MD, Associate Professor,
Section of Neonatology
Sergio Fanella
MD, Associate Professor,
Pediatric Infectious Diseases
sergio.fanella@umanitoba.ca
Department of Pediatrics, Health Sciences Center, University of Manitoba, Canada
The case report
An inborn 25-week male infant weighing 475g was born to a gravida 1 para 0 mother by emergency caesarean section due to uncontrolled blood pressure. The mother had a history of immune thrombocytopenic purpura (ITP) since childhood and the father had factor V Leiden deficiency with a history of pulmonary embolism. There was no family history of frequent or severe infection, stillbirth, unexplained death in infants or young children, or primary immuno-deficiency in the family. Aside from a difficult extraction, the delivery was uncomplicated. The baby’s Apgar scores were 3 and 7 at the first and fifth minute, respectively.
Shortly after delivery he was intubated due to frequent apnoeic events and poor respiratory effort and started on high-frequency jet ventilation, with an initial chest radiograph demonstrating diffuse bilateral granular hazy opacities more marked on the right side. Empiric antibiotics (ampicillin and gentamicin) were started but later discontinued after blood cultures returned negative.
On the third day of life, he developed pancytopenia (white blood cells=1.6x109/L; haemoglobin=118g/L; platelets=40×109/L), which improved with repeated platelet and packed red blood cell transfusions. He continued on high frequency ventilation with settings gradually weaned based on acceptable blood gas results, despite lung imaging showing no improvement. His first trial of extubation at the age of 32 days only lasted for two hours so he was commenced on postnatal steroids using the DART protocol (low dose dexamethasone to facilitate extubation in ventilated babies with evolving or established chronic lung disease).1
He was extubated at the age of 40 days, but was re-intubated one week later due to escalating oxygen requirements while on continuous positive airway pressure (CPAP) therapy with a concurrent rise in his C-reactive protein (CRP) to 105mg/L (normal <5mg/L). A chest radiograph showed extensive air space opacification bilaterally which was worse in the right lung (FIGURE 1).
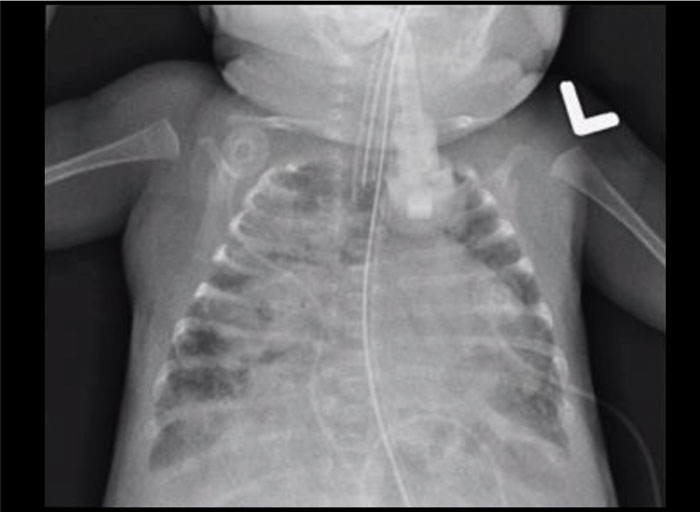
FIGURE 1 Extensive air space opacity mainly on the right lung with patchy opacity in the left lung.
The infant’s blood, urine and cerebro-spinal fluid studies were negative; however, his endotracheal tube culture grew Nocardia farcinica. The infectious diseases and immunology services were consulted. The antibiotics amikacin, meropenem and linezolid were commenced but later changed to oral combination trimethoprim-sulfamethoxazole (TMP-SMX) when susceptibility testing showed that the organism was susceptible to amikacin, amoxicillin-clavulanate and trimethoprim-sulfamethoxazole.
Given that infections with Nocardia are more typically found in immuno-compromised patients, an oxidative burst test was ordered for assessment of chronic granulomatous disease (CGD). Lymphocyte enumeration was normal (CD3=3,226 cells/mm3; CD4=1,843 cells/mm3; CD8=1,267 cells/mm3; CD/CD8 ratio=1.46; CD56=307 cells/mm3; CD19=307 cells/mm3; CD20=307 cells/mm3; CD19CD5=192; CD20CD10≤38). His CD4 recent thymic emigrants were normal (933/mm3). His IgG was low (1.14 g/L; normal=2.61-7.32g/L). His total complement was normal (44.49U/mL). The neutrophil oxidative burst showed a single broad peak that could indicate autosomal recessive CGD.
Regardless of treatment, his radiographs worsened and he developed a requirement of 100% oxygen despite high mean airway pressures of 15-16cmH2O on the high-frequency ventilator. With further increases in mean airway pressure to 18-19cmH2O and prolonged intravenous antibiotic treatment, his radiographs improved. However, his clinical condition eventually deteriorated with an increase in oxygen requirement again to 100% despite attempting different ventilatory strategies and nitric oxide therapy. Based on the inability to wean support, his parents chose to withdraw support at the age of 150 days.
Discussion
Nocardial infection is opportunistic in nature. The majority of infections due to Nocardia species occur in immuno-compromised patients.2 It is caused by ubiquitous aerobic actinomycetes that exist as soil and dust saprophytes.3 The organism, characterised by filamentous branches measuring less than one micrometre thick, is not easily observed on conventional haematoxylin and eosin stained sections, possibly because of its failure to form the ‘granules’ characteristic of other actinomycetes.4 Despite the occurrence of nocardiosis in many mammals (eg cats, dogs, guinea pigs and cattle), there is no evidence of respiratory spread from infected animals to humans.4 The typical portal of entry for Nocardia is the respiratory tract with subsequent dissemination to distant organs or by direct inoculation through the skin. Nosocomial outbreaks of nocardiosis have been reported most frequently for immunocompromised patients.5 None of the reported neonatal cases were linked to in-hospital transmission, with variable ages of presentation between 10 days to two months of age. In our case, the infant was hospitalised from birth until death. What remains unclear in this patient is how he acquired it. A literature review of nosocomial transmission of nocardial infection in heart, liver and renal transplantation units suggested possible transmission via healthcare workers or within the dust and air of the unit.6
Since the 1940s sulfonamides have been the mainstay of therapy. In invasive cases, initial combination therapy should be considered with options including amikacin, a carbapenem, linezolid, cefotaxime and TMP-SMX. Further management is driven by clinical syndrome, species and susceptibility identification, and should be performed in collaboration with an infectious disease consultant.7 Optimal antimicrobial regimens for nocardiosis have not been established due to lack of controlled clinical trials of therapy, lack of firm data on the correlation of in vitro susceptibility with in vivo therapeutic efficacy, and the changing taxonomy of nocardial species.
Chronic granulomatous disease
Although N. asteroides, N. brasiliensis and N. caviae are the main causes of human nocardiosis,8 N. farcinica is a relatively infrequent but clinically aggressive infection, with small numbers of cases reported in immunocompromised patients.8 Nocardia is considered an opportunistic pathogen that may infect individuals with compromised immune function. The relationship between Nocardia species and CGD can be gleaned from an understanding of the immune deficient state of CGD. A retrospective review over 28 years of nocardial infections concluded that Nocardia species are important causes of infection in patients with CGD.9 CGD is a primary immune deficiency that is due to the defective generation of a respiratory burst in human phagocytes (neutrophils, mononuclear cells, macrophages and eosinophils). The resultant defect leads to failure to generate superoxide and an inability to contain certain infectious pathogens.10 There are two subtypes of CGD; X-linked, responsible for the majority of clinical cases, and autosomal recessive forms.
In our infant, the neutrophil oxidative burst showed a single broad peak that may indicate autosomal recessive CGD. Diagnosing CGD in an extremely preterm age group is challenging as the results of oxidative burst testing could be abnormal due to general illness. Moreover, up to one third of nocardial infections occur in individuals without primary immuno-deficiency and chronic lung disease may be a contributing factor in this infection. For these reasons, the infant in this case was to be retested as per the direction from the immunology service, however, the infant died before this could be carried out.
Extreme prematurity as a risk factor for nocardial infection
Extreme prematurity in our neonate could be considered a contributing factor for acquiring nocardial infection. The immune systems of preterm infants have a smaller pool of monocytes and neutrophils, with impaired ability of these cells to kill pathogens. Moreover, lower production of cytokines, which limits T-cell activation, reduces the ability to kill bacteria and detect viruses in cells, compared to term infants. Provision of antenatal corticosteroids has been associated with reductions in lymphocyte proliferation and cytokine production. Invasive medical procedures that are commonplace in the care of these infants, could result in an increased risk of sepsis.11
Conclusion
Nocardia is considered an opportunistic pathogen that is associated with compro-mised immune function. Suppression of cellular immunity and CGD are important risk factors for Nocardia infections. Sulfonamides have been the mainstay of therapy. While an infant born extremely preterm could be at risk of opportunistic infections, the positive test for CGD in this infant and this being the first reported case of N. farcinica in a hospitalised infant, make it highly likely that a primary immunodeficiency was at play. We postulate that, if the innate immune system of a preterm infant puts it at risk of nocardial infections, this would have been established in the literature by now.
Parental consent
The authors received consent to publish this report from the patient’s parents.
Or read this article in our
Tablet/iPad edition
- N. farcinicais an infrequent but clinically aggressive infection usually associated with immunocompromised patients.
- Extreme prematurity may be a contributing factor for acquiring nocardial infection.
Also published in Infant: