Administration of surfactant via the LISA technique in a level 3 unit in Scotland
The LISA (less invasive surfactant administration) procedure delivers surfactant to infants with respiratory distress syndrome via a fine catheter inserted directly into the trachea while maintaining spontaneous respiration on non-invasive ventilation. Following establishment of a standard operating procedure and the training of senior medical staff, the level 3 neonatal unit at Princess Royal Maternity Hospital, Glasgow, was the first in Scotland to implement this procedure. In this article we discuss our procedures and audit our results over a 15-month period.
Asma Yasmeen1,2Neonatal Consultant
asmayasmeen@nhs.net
Anirban Ghosh2
Specialty Registrar Paediatrics
Richard Boulton2
Clinical Scientist
Allan Jackson 2,3
Consultant Neonatologist and ScotSTAR Lead (Neonatal) for Scotland
Carolyn Abernethy2
Consultant Neonatologist
1Victoria Hospital, Kirkcaldy
2Princess Royal Maternity Hospital, Glasgow
3ScotSTAR (Scottish Specialist Transport and Retrieval)
Respiratory distress syndrome (RDS) remains a significant problem following preterm delivery. Surfactant replacement therapy for newborn infants with RDS has been available for about 35 years.1 Current practice is to minimise use of endotracheal ventilation by using less invasive methods for surfactant administration, consequently the LISA technique is gaining popularity around the world.
Evidence on effectiveness of LISA
Analyses of trials to date suggest that the need for mechanical ventilation is reduced in infants where surfactant is administered by the LISA technique. There is also a reduced risk of bronchopulmonary dysplasia (BPD) and death. Because of these benefits, the LISA procedure is recommended in the 2019 European Consensus Guidelines on the management of RDS.2
In a review by Wu et al, the LISA technique was associated with a significant reduction in the risk of mechanical ventilation within 72 hours and BPD at 36 weeks compared to other strategies of surfactant administration.3 These findings were also seen in a meta-analysis by Isayama et al4 and by Aldana-Aguirre et al5 who found a lesser need for mechanical ventilation and a reduction in the composite outcome of death or BPD at 36 weeks compared to the INSURE technique.
Methods – the approach at PRMH
Over the last three to four years, several neonatal units (NNUs) across the UK have embraced the LISA technique.6 Our tertiary NNU at Princess Royal Maternity Hospital (PRMH) was the first in Scotland to implement this procedure. We began training senior medical staff and developed guidelines in 2018.
Stabilisation of preterm infants on continuous positive airway pressure (CPAP) or nasal high flow therapy has become the preferred practice with use of surfactant where required.
Timing of LISA
- In infants <32 weeks’ gestation with a persistent FiO2 (fraction of inhaled oxygen) requirement >30% at any time in the first 72 hours
- In infants ≥32 weeks’ gestation with a persistent FiO2 requirement >40% at any time in the first 72 hours.
Contraindications to performing LISA
Absolute contraindications:
- imminent need for intubation as judged clinically by the attending senior clinician
- maxillofacial, tracheal or known pulmonary malformations
- alternative cause for respiratory distress, eg congenital pneumonia
- no experienced personnel available to perform the LISA procedure.
Relative contraindications:
- severe RDS with high oxygen requirements, severe respiratory acidosis and/or widespread atelectasis on chest X-ray
- infants <26 weeks’ gestation in a unit just starting to use LISA
- pneumothorax requiring drainage
- prominent apnoea, periodic or irregular breathing pattern, despite adequate caffeine citrate administration.
Preparation
We use the Chiesi LISA catheter for the procedure as it was freely available at the time we launched the technique in the department. This catheter is designed to be sited into the trachea via the oral route.
In most cases we use a videolaryngo-scope (Inspiration Healthcare, FIGURE 1). We introduced videolaryngoscopy to the department before initiating the LISA technique and encourage its routine use for endotracheal intubation. We initially trained the most senior staff (consultants and specialty doctors) and then rolled out its use to junior medical staff and advanced neonatal nurse practitioners. During the procedure, the performer uses the live view on the monitor screen attached to the videolaryngoscope as the guide for siting the catheter. Its use has enabled us to safely administer surfactant to the lungs as the introduction of the catheter into the trachea is visible to the whole team. We have found it to be a very useful tool for training junior staff.
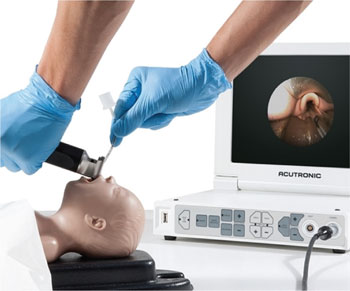
FIGURE 1 The videolaryngoscope (by Inspiration Healthcare).
A LISA ‘pause’ was also established to ensure that the roles and responsibilities of different staff members in the group were clearly assigned before starting the procedure (FIGURE 2).
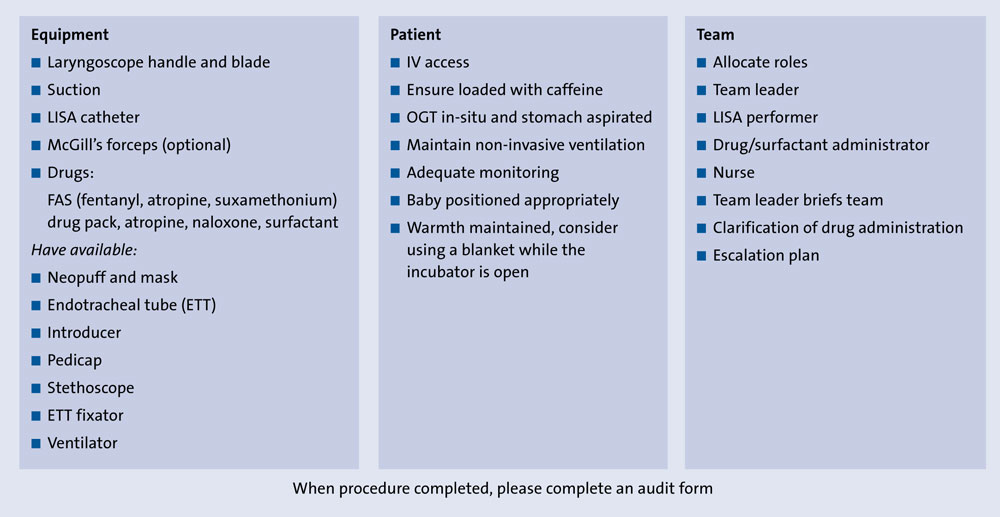
FIGURE 2 The LISA pause.
Awake sedation, other medications and comfort measures
It is known that laryngoscopy can cause adverse physiological responses including systemic, pulmonary and intracranial hypertension and bradycardia, but with the use of pre-medication these effects, as well as pain and discomfort, can be reduced. A recent small randomised control trial by Dekker et al showed there was improved comfort during LISA when propofol (at a dose of 1mg/kg intravenous, IV) was given versus no pre-medication, however there was a slight trend towards more episodes of desaturation and need of non-invasive ventilation with propofol.7 Some give ‘awake’ sedation with fentanyl and keep naloxone available, should there be opiate-induced apnoea or shallow breathing.8 Atropine should also be available for use in instances of prolonged bradycardia. The operator may elect to give atropine prior to the procedure. The protocol for giving medications in our unit is as follows:
- IV fentanyl 1µg/kg given slowly
- have available: atropine 15µg/kg IV; naloxone 10µg/kg IV; a further dose of fentanyl 1µg/kg IV
- a top-up dose of fentanyl to take up to 5µg/kg IV; 2mg/kg suxamethonium IV should also be available in case the infant needs to be intubated and ventilated.
We encourage the routine use of oral sucrose before the LISA procedure in preterm babies due to its analgesic effect. The infant’s comfort is vital for the success of this awake procedure and therefore we also use facilitated tucking.
The procedure
The preparation for LISA is very similar to that for elective intubation. The infant should be positioned as if for intubation and swaddled for comfort. It is important that the baby has continuous monitoring of heart rate and oxygen saturations. They should have an in situ orogastric tube and the stomach should be aspirated. The baby should have intravenous access that has been confirmed to be working. It is important to maintain the baby’s temperature throughout the procedure. Ensure that preterm infants have been loaded with a dose of caffeine citrate given at least 30 minutes prior to the procedure.
A ‘neopuff’ or self-inflating bag and appropriate sized mask should be available as well as appropriately sized endotracheal tubes, should the procedure have to be abandoned due to persistent hypoxaemia, bradycardia or apnoea. Surfactant at 200mg/kg should be drawn into a 5mL syringe with additional air, to be delivered through the cords via a LISA catheter. Where available, the videolaryngoscope should be used to guide the insertion of the LISA catheter through the vocal cords and confirmed by an assistant. Should a videolaryngoscope not be available, or the operator not comfortable or experienced with its use, a standard laryngoscope can be used. In this case, the orogastric tube can be aspirated after the procedure to confirm the delivery of surfactant was not in the stomach.
CPAP or high flow therapy should be maintained throughout the procedure. In some instances, the CPAP prongs may obscure the view for laryngoscopy and the infant may need to be managed on high flow for the duration of the procedure.
Following the administration of IV fentanyl, laryngoscopy is performed and the LISA catheter is inserted through the vocal cords to the desired length: 1.5cm for infants <27 weeks’ gestation and 2cm for infants ≥27 weeks’ gestation. The length of the LISA catheter at the lips is noted to ensure the catheter remains in the same place throughout the procedure. The laryngoscope blade should be gently taken out of the oral cavity, while ensuring that the catheter stays in the same position and the mouth is held closed afterwards. The surfactant in the syringe is then attached to the hub of the LISA catheter and slowly administered over 3-5 minutes by the person assisting the performer. Further slow administration of surfactant might be required if apnoea or bradycardia occurs. Oxygen via CPAP/high flow is titrated to keep oxygen saturation in the normal range. Consider atropine if there is persistent bradycardia and naloxone if apnoeic or shallow breathing despite stimulation. Intermittent positive pressure ventilation might be required briefly in some cases for apnoea. The equipment for endotracheal intubation and ventilation should always be kept ready in case of failed procedure (technical difficulty, prolonged apnoea, bradycardia or hypoxia).
Post-LISA
Following the administration of surfactant, the infants are nursed prone on CPAP. A reduction in oxygen requirement in the next few minutes to hours is taken as indication of improvement and successful outcome. If the infant developes a rapid increase in oxygen requirement, a chest X-ray should be performed to rule out pneumothorax. A further dose of surfac-tant is considered in cases where there is no significant improvement or there is an initial reduction in oxygen requirement and then gradual increase. Some infants require ventilation due to marked work of breathing, a persistently high oxygen requirement, a respiratory acidosis or severe RDS on chest X-ray. In such cases, a further dose of surfactant would be administered via the endotracheal tube.
Review of LISA technique and the effects on long term outcomes
An audit form was devised to gather data for learning and improvement (FIGURE 3). It was completed after every LISA procedure by the person performing the technique.

FIGURE 3 The form for completion after every LISA performance.
We conducted a review of our cases to determine the success rate, untoward effects, BPD rate (<32 weeks’ gestation, 36 weeks’ corrected age) and mortality rate; a case series of all neonates receiving surfactant administration by the LISA technique at PRMH from May 2018 to July 2019. Data were collected from the case report forms, case notes and the electronic patient record database.
Results
A total of 25 infants received surfactant by the LISA technique during the 15-month study period:
- the median gestational age was 30 weeks (range 26-37 weeks)
- the median birth weight was 1,500g (range 690-4,290g)
- there were 17 babies <32 weeks’ gestation
- 16 babies were on nasal CPAP and nine on biphasic CPAP
- the median FiO2 prior to LISA was 35% (range 25-50%)
- the median FiO2 at 60 min post-LISA was 24% (range 21-42%)
- the median surfactant dose was 174mg/kg (range 112-225mg/kg)
- bradycardia of <80 beats/min was observed in 11 cases during the procedure
- endotracheal intubation and mechanical ventilation was required in seven (28%) cases within 48 hours post-LISA
- there were no cases of pneumothorax or significant intraventricular haemorrhage (grade 3 or more) post procedure
- no deaths were recorded in the LISA cohort.
Staff
The technique of LISA is very similar to endotracheal intubation and staff trained in endotracheal intubation were able to easily adapt to this procedure. No attempts were abandoned or changed to intubation because of technical difficulty. The LISA procedure was successfully performed in all cases where attempted (within three attempts). The operator of the procedure was a consultant in 17 cases, specialty doctor in six cases and a neonatal registrar (ST3-ST8) in two cases.
Medication
We also studied the use of different medications:
- pre-procedure fentanyl was given in 24 cases
- oral sucrose use was documented in only two cases
- atropine was used in eight cases (pre-procedure or post-bradycardia during the procedure)
- naloxone was only required in one case.
Mechanical ventilation
The characteristics of infants who required mechanical ventilation (n=7) were compared to those who did not. Both groups had a median gestation of 30 weeks’ with a range of 26-36 weeks’ in the intubated group and 27-37 weeks’ in the group who did not require intubation. Median birth weight was lower in the intubated group (1,330g; range=690-2,786g) compared to the non-intubated group (1,530g; range=1,010-4,290g). The median FiO2 pre-procedure was higher in the intubated group (40%; range=32-50%) than in the non-intubated group (35%; range= 25-50%).
Overall, the BPD rate for babies born at <32 weeks’ gestation (n=80) was 38.7% during the study period. In babies <32 weeks’ gestation receiving surfactant via the LISA technique (n=17), the BPD rate was 17.6%. This might indicate that the sicker babies were ventilated and progressed to have BPD relatively more frequently than the infants who were clinically better and were given LISA. It might also indicate beneficial effects of LISA on reduction of BPD. Given that our numbers are small and it was not a randomised controlled trial, the results need to be interpreted with caution.
There were no deaths recorded among the infants who received surfactant via the LISA technique. There were a total of five deaths among all babies born at <32 weeks’ gestation (n=80) in the unit during the study period.
Conclusions
We have successfully introduced the LISA technique into our department. No significant adverse effects have been seen to date and the procedure has been successful in all babies in which it was attempted. Transient bradycardia was frequently observed, which generally resolved with minimal intervention. Seventy-two per cent of the patients in our series avoided intubation and ventilation. Intubated babies tended to be smaller, less mature and requiring more oxygen. A trend towards reduction in BPD rates was seen, although we acknowledge the small sample size. Further work is required on patient selection, timing of the procedure and optimal medications.
Or read this article in our
Tablet/iPad edition
- Structured guidelines and training of medical staff are required to establish LISA as a routine procedure in the NNU.
- Videolaryngoscopy is a useful adjunct to the procedure.
- Further studies are required on the use of medications during the procedure.
- A robust system of data collection and analysis helps to guide improvements.